What is PML and what causes it?
Progressive multifocal leukoencephalopathy (PML) is a rare opportunistic brain infection that is caused by the human polyomavirus JC virus (JCV). The prevalence of JCV in healthy blood donors and in patients with multiple sclerosis (MS) is at least 50% and up to 80% in older cohorts.1-3 It is transmissible via the respiratory tract and fecal-oral.3 The virus can be latent and persist in the kidney, bone marrow and lymphoid tissue in asymptomatic immunocompetent individuals within precursor B-cells, CD34+ hematopoietic progenitor cells and tonsillar stromal cells.3 However, in immunocompromised patients, including those taking certain MS disease-modifying therapies (DMTs), JCV replicates more and accumulates complex errors that facilitate enhanced entry into the central nervous system (CNS), replication efficiency and cell-specific tropism for oligodendrocytes, leading to their lysis and subsequent demyelination. Host genetic risk factors (e.g. HLA-DRB1*0401 allele and p53 polymorphisms) can also increase susceptibility for PML. The precise mechanism of CNS entry is unknown. The spectrum of JCV infection includes demyelinating lesions with lytic infection of oligodendrocytes. PML variants that infect neurons are seen with cerebellar granule cell neuronopathy (progressive cerebellar atrophy) and encephalopathy (lytic infection of cortical pyramidal neurons leading to pronounced cortical atrophy). Infection of leptomeninges and choroid plexus has also been reported.4-7 The JCV found in the CNS during PML is a mutated, neurotropic variant distinct from the latent archetype in the kidneys and exhibits 100–1,000-fold higher viral loads in infected brain cells. In contrast, the archetype is shed only intermittently in urine and is difficult to detect without an indirect, stratified JCV assay.
When should a clinician suspect PML in a person with MS?
A new or enlarging MRI brain lesion and/or progressive neurologic symptoms in the setting of immunosuppressive medications, particularly the α4 integrin blocker, natalizumab, should raise concern for PML. Natalizumab exposure for more than 2 years in an individual who is JCV seropositive carries a particularly greater risk for PML. PML has also been rarely seen with other DMTs (e.g. fingolimod, Siponimod, ozanimod, dimethyl fumarate, cladribineinterferon beta-1a and ocrelizumab). Radiographic changes include new or enlarging T2/FLAIR lesions, paramagnetic rims at the gray-white matter junction of new/enlarging juxtacortical lesions, and rim gadolinium enhancement. Clinically, symptoms can vary from weakness, paresthesias, cognitive or behavioral changes, gait dysfunction, speech/language difficulties, visual field defects or seizure. These symptoms can be insidious in patients with MS on DMTs relative to those with PML due to HIV. However, PML can be asymptomatic for many months prior to clinical presentation and observed with new lesions on MRI. Patients should be monitored closely via MRI and undergo lumbar puncture for JCV PCR if there is a suspicion for PML. See Table 1 for further diagnostic details.
How is PML diagnosed?
Integration of imaging, clinical and laboratory evidence is useful in the diagnosis of PML (Table 1).2
PML encompasses a spectrum with an absent antiviral immune response on one end (in the setting of a severely immunocompromised state, e.g. AIDS, hematological malignancy), inflammatory PML in the middle (with some preserved immune function), to extensive inflammation in the setting of immune reconstitution (IRIS). The latter two presentations are seen in patients with MS.
Brain MRI can demonstrate one or more T2/FLAIR hyperintense and T1 hypointense lesions involving the subcortical and juxtacortical white matter (Figure 1). Up to half of patients on MS DMT-associated PML exhibit faint gadolinium rim enhancement, so this is not uncommon at the time of PML diagnosis.2, 6 PML lesions can be multifocal or unifocal. The dynamic nature of MRI findings can contribute to diagnosis, since PML becomes unlikely if the MRI manifestations are stable over weeks to a few months on serial imaging (Figure 1). Asymptomatic lesions are typically focal (68%) and in the frontal lobe (78%).3 Clinical involvement of the optic nerve or spinal cord has not been reported with PML. Repeating an MRI brain in 1 month and then 3 months to evaluate for interval change will be useful.
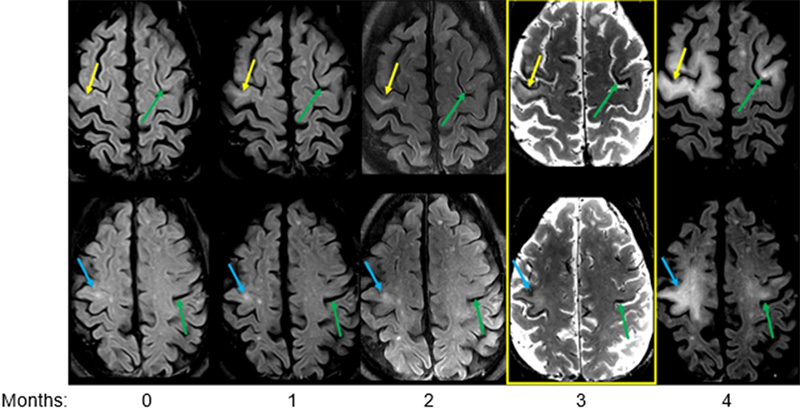
Figure 1. Progression of PML lesions on MRI brain. Progression of PML lesions (each colored arrow represents a unique lesion) on 1.5 T brain MRI from left to right through time (months: 0–4). Highlighted panel in yellow marking 7 T SWI with a hypointense band along the cortical-WM boundary corresponding to FLAIR hyperintensity. FLAIR hyperintensities are subtle and months 0–2 preceding acquisition of the SWI sequence. The FLAIR hyperintensities associated with the SWI changes continue to evolve and become more confluent from months 3 to 4. Susceptibility changes correspond to iron accumulation in macrophages at the gray-white matter junction (see manuscript for details8).
Immediate evaluation with lumbar puncture for CSF JCV PCR is reasonable depending on the patient’s JCV antibody status and clinical history. To improve the sensitivity of detecting JCV, as viral titers can be low and difficult to identify in the CSF, specimens from our center are sent to Focus Laboratories Material Research Laboratories (MRL) for ultrasensitive (lower limit of quantification [LLOQ] 10 copies/mL) quantitative real-time PCR testing. Ensuring the JCV antibody assay performed by a lab has an LLOQ of 10 copies/mL is recommended.
While serum JCV antibody testing is useful in assessing the risk of PML, it does not indicate the presence or absence of PML. In patients who are JCV antibody positive, shared decision making with the JCV antibody titer (e.g. STRATIFY antibody index) can be done to determine the risk of PML (Table #). JCV is only sporadically shed in the urine; we do not recommend JCV PCR.
Although diagnosis can generally be established via clinical history, MRI and CSF analysis, brain biopsy is sometimes obtained when the diagnosis of PML has not been confirmed. Prior to biopsy, it is worthwhile to repeat the CSF sampling at least once unless the rate of progression suggests the need to immediately sample tissue to find an alternative diagnosis. Histopathological examination of brain tissue confirming PML will demonstrate characteristic demyelination, bizarre astrocytes with atypical nuclei, perivascular cuffing and oligodendroglial nuclear inclusions seen with PML.9 These findings should be confirmed with immunohistochemistry for the viral capsid protein VP1 or tissue JCV PCR.
| Modality | Findings |
|---|---|
| Clinical history | Subacute (insidious) onset of weakness, paresthesias, cognitive or behavioral changes, gait dysfunction, speech/language difficulties or seizure; can be asymptomatic early. |
| MRI brain | ≥1 T2/FLAIR hyperintense and T1 hypointense or paramagnetic changes involving the juxtacortical white matter; faint rim enhancement. |
| CSF | Ultrasensitive JCV PCR, lower limit of quantification 10 copies/mL |
| Brain biopsy | Demyelination, oligodendroglia with enlarged hyperchromatic nuclei, enlarged bizarre astrocytes with irregularly lobulated nuclei, and oligodendroglial nuclear inclusions; viral particles (VP1 capsid) in oligodendrocytes, astrocytes or neurons; JCV PCR. |
Table 1. Evidence supporting the diagnosis of PML.
What are mimics of PML that should be considered in this evaluation?
In the process of evaluating a patient with suspected PML, other etiologies to consider include posterior reversible encephalopathy syndrome (PRES), CNS vasculitis, VZV leukoencephalitis, neoplasm (metastases, glioma and lymphoma), HSV encephalitis (especially in the setting of seizure and temporal lobe lesions), autoimmune encephalitis and MS disease activity.10
What is immune reconstitution inflammatory syndrome (IRIS)?
Although immune reconstitution from withdrawal of immunosuppression is desired for management of PML and results in better outcomes, immune reconstitution inflammatory syndrome (IRIS) can be life-threatening if the inflammatory response to the newly recognized JCV CNS infection is extensive. MRI changes include contrast enhancement within the PML lesion, edema and mass effect. Pathologically, perivascular lymphocytic infiltrates comprised of predominantly CD8+ T-cells; CD20+ B-cells, CD138+ plasma cells, microglia and macrophages are observed. IRIS represents a paradox where immune reconstitution is necessary for viral control, but an overly robust inflammatory response against JCV-infected CNS tissue is deleterious.
Why is natalizumab associated with a higher risk of PML in MS?
Natalizumab may mediate mobilization of bone marrow precursor CD34+ hematopoietic and CD19+ B-cells to facilitate JCV reactivation and dissemination. Natalizumab blocks α4-integrin–mediated adhesion of lymphocytes to VCAM-1 on CNS endothelium, thereby inhibiting trafficking of JCV-specific CD4⁺ and CD8⁺ T cells into the brain. This limits immune surveillance and allows uncontrolled JCV replication in glial cells, leading to PML.
In those with negative JCV titers, the incidence of PML with Tysabri use is 1/10,000.
With positive JCV titers, treatment duration is associated with increased risk of PML.
Although JCV titers may fluctuate and rarely revert to negative, a history of JCV positivity is usually interpreted as evidence of prior exposure.
PML has also been observed with extended interval dosing of natalizumab, although the risk may be lower (0.08 cases/100 persons with extended dosing versus 0.3 cases/100 persons with standard dosing).11, 12
Four rare deleterious germline alleles associated with PML have been identified that confer an increased risk of PML in patients exposed to PML-associated drugs. Despite a commercially available test for these variants existing, we do not currently utilize this testing in our clinical practice.13
To mitigate these risks, we favor alternatives to natalizumab in treating MS in people who are JCV seropositive.
| Natalizumab duration (months) | Risk of PML
Prior use of immunosuppressants* |
|
|---|---|---|
| No | Yes | |
| 1-24 | < 1/1,000 | 1/1,000 |
| 25-48 | 2/1,000 | 6/1,000 |
| 49-72 | 4/1,000 | 7/1,000 |
| 73-96 | 2/1,000 | 6/1,000 |
Table 2. Risk of PML with prior use of immunosuppressants by natalizumab exposure and STRATIFY JCV antibody index.14
*(e.g. 6-mercaptopurine, cyclosporine, methotrexate, azathioprine, TNF-α inhibitors or chronic steroid use)15
| Natalizumab duration (months) | Risk of PML Without prior immunosuppressants* |
Risk of PML
With prior immunosuppressants* |
|||
| No index value | Index <0.9 | Index <0.9 | Index >1.5 | ||
| 1-12 | 1/10,000 | 1/10,000 | 1/10,000 | 1/5,000 | 1/3,333 |
| 13-24 | 1/1,667 | 1/10,000 | 1/3,333 | 1/1,111 | 1/2,500 |
| 25-36 | 1/500 | 1/5,000 | 1/1,250 | 1/333 | 1/250 |
| 37-48 | 1/250 | 1/2,500 | 1/500 | 1/142 | 1/125 |
| 49-60 | 1/200 | 1/2,000 | 1/500 | 1/125 | 1/125 |
| 61-72 | 1/167 | 1/1,667 | 1/333 | 1/100 | 1/167 |
Table 3. Risk of PML by STRATIFY JCV antibody index tiers16
*(e.g. 6-mercaptopurine, cyclosporine, methotrexate, azathioprine, TNF-α inhibitors or chronic steroid use)15
Which other DMTs have been associated with PML (in MS and non-MS cases)?
Cases of PML have been reported with the use of other MS DMTs, both independent of and following use of natalizumab.
In a cohort of 57 cases of PML in patients with MS on sphingosine-1-phosphate receptor modulators (S1PRm), 53 were while on fingolimod.17 PML has also been reported with siponimod.18 An asymptomatic case of PML-IRIS identified with MRI and elevated serum neurofilament light levels was reported with ozanimod.19
21 reported cases of PML associated with dimethyl fumarate occurred with absolute lymphocyte counts (ALC) less than 800 cells/mm.20 In another study, older age and persistent lymphopenia (ALC < 800) were associated with PML.21 An overexpression of PD-1 on CD8+ and memory effector T-cells was found with preserved lymphocyte counts in a case of PML with dimethyl fumarate. 320
Two cases of PML associated with cladribine in individuals without MS have been reported.22, 23
Two cases of PML associated with current and remote exposure to interferon beta-1a have been reported.24, 25
A single case of PML and another with IRIS without clinical PML have been reported with alemtuzumab.26, 27 Other cases of alemtuzumab associated with PML in non-MS individuals have been reported.28-30
The incidence of PML with rituximab is 1/10,000 from a meta-analysis of 10 studies.12 A case of PML with ocrelizumab has been reported.19 A case of PML in an individual with chronic lymphocytic leukemia on ofatumumab was reported.31
A case of PML following anti-CD19-directed CAR T therapy for diffuse large B-cell lymphoma has been reported.32
What are treatment strategies for treating PML?
The offending DMT should be immediately discontinued.
With teriflunomide, we recommend the use of cholestyramine to accelerate drug clearance.
No adjunctive therapy has been demonstrated to be effective in a randomized controlled trial.33
Plasmapheresis (plasma exchange, PLEX) for five 1.5-volume exchanges, performed every other day can be considered, particularly if a monoclonal antibody has been administered in the past 2-3 months and the clinical presentation of PML is severe. With natalizumab, we have recommended PLEX to accelerate its clearance and restore peripheral lymphocyte transmigration to the CNS across the blood-brain barrier.9, 34 The clinical benefit of PLEX is unclear, and it may be associated with more severe IRIS.35-38
The checkpoint inhibitor program cell death-1 (PD-1) has been shown to be over-expressed on CD4+ and CD8+ T-cells in patients with PML and can be considered.39 Pembrolizumab 200 mg every 3 weeks for 5 infusions) has been reported to be useful in a case of natalizumab-associated PML.40 A case of fingolimod-associated PML has also been treated with pembrolizumab 2 mg/kg IV monthly for 3 doses.41 Prospective open-label studies have had mixed results of improvement/ stabilization, no improvement/death.42, 43 Two cases of PML in patients without MS or HIV did not show benefit with pembrolizumab.44
Another PD-1 inhibitor, nivolumab 1-3 mg/kg every 2 weeks for 2 doses, followed by 3mg/kg for the third dose, can also be considered.43
Consultation with hematology/oncology is recommended for using PD-1 inhibitors.
Intravenous immunoglobulin (IVIg) can also be considered in the setting of hypogammaglobulinemia.45-47
Attempting to inhibit viral entry into glial cells via the 5-HT2A serotonin receptor with mirtazapine has not demonstrated benefit.48
The antimalarial mefloquine, which inhibits viral infection and replication in vitro, has also been evaluated without convincing benefit and has potential psychotropic side-effects; we do not recommend its use.
Filgrastim (GM-CSF) has been used with some positive results; we do not currently recommend its use as it may increase the risk of IRIS and has modest, if any, benefit.49-51
Table 4 summarizes treatment options.
How is PML-related IRIS managed?
IRIS develops when the immune system recognizes PML infection and mounts an inflammatory response. While HIV PML-IRIS has been associated with better prognosis, early IRIS with natalizumab-associated PML was associated with worse survival and neurological outcome. 37, 52 Generally, over-exuberant IRIS is managed with intravenous methylprednisolone to suppress the overactive immune response. Given the need for immune reconstitution for control of JCV infection, the balance between immune response against JCV infection and immunosuppression to moderate IRIS can be challenging.53
If a patient with PML develops significantly disruptive clinical symptoms of IRIS and MRI confirms inflammation (i.e. interval increase in T2 hyperintensities typically with gadolinium enhancement), methylprednisolone 1 g/day IV for 3-5 days should be administered. Periodic clinical and MRI reassessment should be performed to follow the response to therapy, and methylprednisolone IV can be repeated every 4 weeks if needed to control severe clinical manifestations of IRIS. Isolated MRI changes without clinical worsening do not necessarily warrant change in management. In practice, balancing tolerable IRIS against excessive inflammation requires using judgement based on clinical and radiological findings.
Maraviroc, a CCR5 chemokine receptor antagonist, is hypothesized to decrease excessive recruitment of CCR5+ T-cells into the CNS, driving IRIS.53, 54 The clinical effectiveness of maraviroc has not been established, and results have been mixed.55-58 Potential side effects can be severe and include hepatotoxicity, infections and skin hypersensitivity reactions.
Additional treatments in Table 4 can be considered.
| Therapy | Proposed Mechanism | Dose |
| PLEX | Accelerate clearance of the therapy to expedite immune reconstitution. | 5 exchanges of 1.5x plasma-volume performed every other day |
| Pembrolizumab | Inhibits PD-1 on JCV-specific T-cells to facilitate clearance of JCV-infected cells. | 2 mg/kg or 200 mg every 3-4 weeks for 3-5 doses |
| Nivolumab | Inhibits PD-1 on JCV-specific T-cells to facilitate clearance of JCV-infected cells. | 1-3 mg/kg every 2 weeks for 2 doses, followed by 3mg/kg for 3rd dose |
| Intravenous immunoglobulin | Provide passive humoral immunity in setting of hypogammaglobulinemia. | 0.4 g/kg/day for 5 days |
| Mirtazapine | Inhibits JCV entry into glia via the 5-HT2A receptor, thereby reducing replication and cell lysis. | 30 to 45mg daily, with a maximum dosage of 60 mg daily |
| Maraviroc | Reduce recruitment of CCR5+ lymphocytes to CNS during IRIS. | 300 mg twice a day |
Table 4. Potential interventions in management of PML/IRIS
What are the common complications of PML?
Depression is commonly seen in patients who develop PML and should be treated with antidepressants and psychotherapy. Given the potential antiviral benefit of mirtazapine, this is often considered a first-line treatment of depression. Seizures are occasionally seen, although they can be subtle, particularly if they arise from the frontal or temporal lobes. We would start antiseizure medications after a seizure has occurred.
What is the prognosis of PML secondary to MS therapies?
Earlier diagnosis is associated with better outcomes from PML. The ability to survive the initial PML infection and safely navigate the immune reconstitution inflammatory response is associated with variable but potentially good long-term outcomes. In general, residual neurological deficits are common. In patients with natalizumab-associated PML, survival is estimated to be 96% in asymptomatic and 75% in symptomatic individuals.58 Factors associated with better prognosis include younger age at diagnosis, lower pre-PML disability, and early recognition and diagnosis. Clinical stability typically takes 3-6 months after the diagnosis of PML is made, with continued improvement possible over the subsequent 6-12 months.59
Viral persistence in CSF of the mutated JCV following PML infection and immune reconstruction can be detected for years, even in a clinically stable patient. Therefore, repeat CSF testing is not indicated since it does not impact long-term course or management. Risk of recurrence is unknown but has been reported in patients with HIV and CLL, where long-term immune reconstitution is more challenging than MS. 60, 61 At the Mellen Center, we have several MS patients who remain clinically stable 14+ years after diagnosis of PML. Clinical and radiographic surveillance for return of MS disease-activity PML is recommended. If necessary, restarting DMTs with lower-efficacy DMTs is reasonable to consider, but typically only after demonstrated return of MS disease activity. A cohort of 23 people with MS who developed natalizumab-PML were restarted on low or moderate efficacy DMTs (interferons, S1PRm, dimethyl fumarate and glatiramer acetate) after PML resolution did not develop recurrence or clinical worsening (mean follow-up 24.1 months).62 Switching to teriflunomide may also be reasonable, as some data suggests it may inhibit JCV spread.63
References
- Egli A, Infanti L, Dumoulin A, Buser A, Samaridis J, Stebler C, Gosert R, Hirsch HH. Prevalence of polyomavirus BK and JC infection and replication in 400 healthy blood donors. J Infect Dis. 2009;199(6):837-46. doi: 10.1086/597126. PubMed PMID: 19434930.
- Bozic C, Subramanyam M, Richman S, Plavina T, Zhang A, Ticho B. Anti-JC virus (JCV) antibody prevalence in the JCV Epidemiology in MS (JEMS) trial. Eur J Neurol. 2014;21(2):299-304. Epub 20131130. doi: 10.1111/ene.12304. PubMed PMID: 24894998.
- Cortese I, Reich DS, Nath A. Progressive multifocal leukoencephalopathy and the spectrum of JC virus-related disease. Nat Rev Neurol. 2021;17(1):37-51. Epub 20201120. doi: 10.1038/s41582-020-00427-y. PubMed PMID: 33219338; PMCID: PMC7678594.
- Reoma LB, Trindade CJ, Monaco MC, Solis J, Montojo MG, Vu P, Johnson K, Beck E, Nair G, Khan OI, Quezado M, Hewitt SM, Reich DS, Childs R, Nath A. Fatal encephalopathy with wild-type JC virus and ruxolitinib therapy. Ann Neurol. 2019;86(6):878-84. Epub 20191016. doi: 10.1002/ana.25608. PubMed PMID: 31600832; PMCID: PMC8189164.
- Agnihotri SP, Wuthrich C, Dang X, Nauen D, Karimi R, Viscidi R, Bord E, Batson S, Troncoso J, Koralnik IJ. A fatal case of JC virus meningitis presenting with hydrocephalus in a human immunodeficiency virus-seronegative patient. Ann Neurol. 2014;76(1):140-7. Epub 20140620. doi: 10.1002/ana.24192. PubMed PMID: 24895208; PMCID: PMC4112354.
- Tan CS, Koralnik IJ. Progressive multifocal leukoencephalopathy and other disorders caused by JC virus: clinical features and pathogenesis. Lancet Neurol. 2010;9(4):425-37. doi: 10.1016/S1474-4422(10)70040-5. PubMed PMID: 20298966; PMCID: PMC2880524.
- Schippling S, Kempf C, Buchele F, Jelcic I, Bozinov O, Bont A, Linnebank M, Sospedra M, Weller M, Budka H, Martin R. JC virus granule cell neuronopathy and GCN-IRIS under natalizumab treatment. Ann Neurol. 2013;74(4):622-6. Epub 20130916. doi: 10.1002/ana.23973. PubMed PMID: 23868420.
- Mahajan KR, Amin M, Poturalski M, Lee J, Herman D, Zheng Y, Androjna C, Howell M, Fox RJ, Trapp BD, Jones SE, Nakamura K, Ontaneda D. Juxtacortical susceptibility changes in progressive multifocal leukoencephalopathy at the gray-white matter junction correlates with iron-enriched macrophages. Mult Scler. 2021;27(14):2159-69. Epub 20210322. doi: 10.1177/1352458521999651. PubMed PMID: 33749379; PMCID: PMC8455719.
- von Einsiedel RW, Fife TD, Aksamit AJ, Cornford ME, Secor DL, Tomiyasu U, Itabashi HH, Vinters HV. Progressive multifocal leukoencephalopathy in AIDS: a clinicopathologic study and review of the literature. J Neurol. 1993;240(7):391-406. doi: 10.1007/BF00867351. PubMed PMID: 8410079.
- Berger JR, Aksamit AJ, Clifford DB, Davis L, Koralnik IJ, Sejvar JJ, Bartt R, Major EO, Nath A. PML diagnostic criteria: consensus statement from the AAN Neuroinfectious Disease Section. Neurology. 2013;80(15):1430-8. doi: 10.1212/WNL.0b013e31828c2fa1. PubMed PMID: 23568998; PMCID: PMC3662270.
- Takahashi K, Nakahara J, Miura Y, Ae R, Nakamichi K, Harada M, Mori K, Sanjo N, Yukitake M, Yokote H, Hamaguchi T, Yamada M, Takao M. Natalizumab-Associated Progressive Multifocal Leukoencephalopathy After Natalizumab Extended Interval Dosing Therapy in Japan. Neurol Neuroimmunol Neuroinflamm. 2025;12(6):e200476. Epub 20250828. doi: 10.1212/NXI.0000000000200476. PubMed PMID: 40875960; PMCID: PMC12396746.
- Rindi LV, Zace D, Braccialarghe N, Massa B, Barchi V, Iannazzo R, Fato I, De Maria F, Kontogiannis D, Malagnino V, Sarmati L, Iannetta M. Drug-Induced Progressive Multifocal Leukoencephalopathy (PML): A Systematic Review and Meta-Analysis. Drug Saf. 2024;47(4):333-54. Epub 20240207. doi: 10.1007/s40264-023-01383-4. PubMed PMID: 38321317.
- Hatchwell E, Smith EB, 3rd, Jalilzadeh S, Bruno CD, Taoufik Y, Hendel-Chavez H, Liblau R, Brassat D, Martin-Blondel G, Wiendl H, Schwab N, Cortese I, Monaco MC, Imberti L, Capra R, Oksenberg JR, Gasnault J, Stankoff B, Richmond TA, Rancour DM, Koralnik IJ, Hanson BA, Major EO, Chow CR, Eis PS. Progressive multifocal leukoencephalopathy genetic risk variants for pharmacovigilance of immunosuppressant therapies. Front Neurol. 2022;13:1016377. Epub 20221214. doi: 10.3389/fneur.2022.1016377. PubMed PMID: 36588876; PMCID: PMC9795231.
- Biogen. [11/6/25]. Available from: https://www.tysabrihcp.com/en_us/home/efficacy-safety/pml-risk.html.
- (FDA) UFDA. Tysabri FDA Full Prescribing Information [updated 3/20251/5/26]. Available from: https://www.accessdata.fda.gov/spl/data/119c87a9-d153-4796-9525-694afdcdc1d6/119c87a9-d153-4796-9525-694afdcdc1d6.xml.
- BiogenLinc. PML tools and StratifyJCV [updated 2/20251/5/26]. Available from: https://biogenlinc.co.uk/en/products/ms-portfolio/tysabri/pml/.
- Croteau D, Kim T, Chan V, Stevens J, Pimentel Maldonado DA, Baldassari LE, Lee PR, Hughes A, Brinker A. Progressive multifocal leukoencephalopathy associated with sphingosine-1-phosphate receptor modulators: A large case series. Mult Scler Relat Disord. 2024;92:106163. Epub 20241106. doi: 10.1016/j.msard.2024.106163. PubMed PMID: 39541823.
- Rot U, Jerala M, Horvat Ledinek A, Brecl Jakob G. Progressive multifocal leukoencephalopathy in association with siponimod treatment for secondary progressive multiple sclerosis: a case report. J Neurol. 2024;271(9):6363-7. Epub 20240703. doi: 10.1007/s00415-024-12542-7. PubMed PMID: 38960947.
- Quintanilla-Bordas C, Gorriz D, Cubas-Nunez L, Castillo-Villalba J, Carreres-Polo J, Casanova B, Perez-Miralles FC. Elevation of serum neurofilament light-chain levels disclose possible occult progressive multifocal leukoencephalopathy and immune reconstitution syndrome in a patient receiving ozanimod: a case report. Front Immunol. 2024;15:1465678. Epub 20241022. doi: 10.3389/fimmu.2024.1465678. PubMed PMID: 39502687; PMCID: PMC11535851.
- Garcia J, Hendel-Chavez H, De-Goer MG, L'Honneur AS, Dubessy AL, Taoufik Y, Stankoff B. Progressive multifocal leukoencephalopathy on dimethyl fumarate with preserved lymphocyte count but deep T-cells exhaustion. Mult Scler. 2021;27(4):640-4. Epub 20200720. doi: 10.1177/1352458520942201. PubMed PMID: 32686582.
- Jordan AL, Yang J, Fisher CJ, Racke MK, Mao-Draayer Y. Progressive multifocal leukoencephalopathy in dimethyl fumarate-treated multiple sclerosis patients. Mult Scler. 2022;28(1):7-15. Epub 20200818. doi: 10.1177/1352458520949158. PubMed PMID: 32808554; PMCID: PMC7889744.
- Berghoff M, Schanzer A, Hildebrandt GC, Dassinger B, Klappstein G, Kaps M, Gizewski ER, Acker T, Grams A. Development of progressive multifocal leukoencephalopathy in a patient with non-Hodgkin lymphoma 13 years after treatment with cladribine. Leuk Lymphoma. 2013;54(6):1340-2. Epub 20121120. doi: 10.3109/10428194.2012.740669. PubMed PMID: 23098217.
- Alstadhaug KB, Fykse Halstensen R, Odeh F. Progressive multifocal leukoencephalopathy in a patient with systemic mastocytosis treated with cladribine. J Clin Virol. 2017;88:17-20. Epub 20161214. doi: 10.1016/j.jcv.2016.12.005. PubMed PMID: 28095350.
- Lehmann HC, Kruger K, Fink GR, Schroeter M. Progressive multifocal leukoencephalopathy after interferon beta-1a monotherapy. J Neurol. 2015;262(3):771-3. Epub 20150113. doi: 10.1007/s00415-014-7620-4. PubMed PMID: 25578178.
- Gonzalez SC, Nguyen A, Soto JM, Shan Y. Ring-Enhancing Progressive Multifocal Leukoencephalopathy Mimicking Glioma in a Presumed Immunocompetent Patient With a History of Multiple Sclerosis: A Case Report and Review of the Literature. Cureus. 2023;15(9):e45543. Epub 20230919. doi: 10.7759/cureus.45543. PubMed PMID: 37868479; PMCID: PMC10585186.
- Jamroz-Wisniewska A, Drelich-Zbroja A, Chyrchel-Paszkiewicz U, Rejdak K. IRIS without PML in MS patients - a case report of IRIS after past alemtuzumab therapy with subsequent oral cladribine treatment and review of the literature. Ann Agric Environ Med. 2025;32(3):428-31. Epub 20250217. doi: 10.26444/aaem/200751. PubMed PMID: 41025192.
- Gerevini S, Capra R, Bertoli D, Sottini A, Imberti L. Immune profiling of a patient with alemtuzumab-associated progressive multifocal leukoencephalopathy. Mult Scler. 2019;25(8):1196-201. Epub 20190409. doi: 10.1177/1352458519832259. PubMed PMID: 30964396.
- Keene DL, Legare C, Taylor E, Gallivan J, Cawthorn GM, Vu D. Monoclonal antibodies and progressive multifocal leukoencephalopathy. Can J Neurol Sci. 2011;38(4):565-71. doi: 10.1017/s0317167100012105. PubMed PMID: 21672696.
- Isidoro L, Pires P, Rito L, Cordeiro G. Progressive multifocal leukoencephalopathy in a patient with chronic lymphocytic leukaemia treated with alemtuzumab. BMJ Case Rep. 2014;2014. Epub 20140108. doi: 10.1136/bcr-2013-201781. PubMed PMID: 24403383; PMCID: PMC3902436.
- Avila JD. Alexia Without Agraphia as the Initial Manifestation of Progressive Multifocal Leukoencephalopathy in Chronic Lymphocytic Leukemia. Neurohospitalist. 2020;10(1):71-2. Epub 20190410. doi: 10.1177/1941874419840900. PubMed PMID: 31839871; PMCID: PMC6900653.
- Hashimoto Y, Tashiro T, Ogawa R, Nakamichi K, Saijo M, Tateishi T. Therapeutic Experience of Progressive Multifocal Leukoencephalopathy Development during Ofatumumab Therapy for Chronic Lymphocytic Leukemia. Intern Med. 2021;60(24):3991-3. Epub 20210626. doi: 10.2169/internalmedicine.6723-20. PubMed PMID: 34176829; PMCID: PMC8758456.
- Sdrimas K, Diaz-Paez M, Camargo JF, Lekakis LJ. Progressive multifocal leukoencephalopathy after CAR T therapy. Int J Hematol. 2020;112(1):118-21. Epub 20200303. doi: 10.1007/s12185-020-02840-x. PubMed PMID: 32125605.
- Glenn T, Berger JR, McEntire CRS. Natalizumab-associated progressive multifocal leukoencephalopathy. Front Neurol. 2025;16:1575653. Epub 20250620. doi: 10.3389/fneur.2025.1575653. PubMed PMID: 40621097; PMCID: PMC12226301.
- Khatri BO, Man S, Giovannoni G, Koo AP, Lee JC, Tucky B, Lynn F, Jurgensen S, Woodworth J, Goelz S, Duda PW, Panzara MA, Ransohoff RM, Fox RJ. Effect of plasma exchange in accelerating natalizumab clearance and restoring leukocyte function. Neurology. 2009;72(5):402-9. doi: 10.1212/01.wnl.0000341766.59028.9d. PubMed PMID: 19188571; PMCID: PMC2677532.
- Landi D, De Rossi N, Zagaglia S, Scarpazza C, Prosperini L, Albanese M, Buttari F, Mori F, Marfia GA, Sormani MP, Capra R, Centonze D, Italian PMLsg. No evidence of beneficial effects of plasmapheresis in natalizumab-associated PML. Neurology. 2017;88(12):1144-52. Epub 20170222. doi: 10.1212/WNL.0000000000003740. PubMed PMID: 28228569.
- Scarpazza C, Prosperini L, De Rossi N, Moiola L, Sormani MP, Gerevini S, Capra R, Italian PMLg. To do or not to do? plasma exchange and timing of steroid administration in progressive multifocal leukoencephalopathy. Ann Neurol. 2017;82(5):697-705. Epub 20171031. doi: 10.1002/ana.25070. PubMed PMID: 29023856.
- Tan IL, McArthur JC, Clifford DB, Major EO, Nath A. Immune reconstitution inflammatory syndrome in natalizumab-associated PML. Neurology. 2011;77(11):1061-7. Epub 20110810. doi: 10.1212/WNL.0b013e31822e55e7. PubMed PMID: 21832229; PMCID: PMC3174071.
- Tyler KL, Vollmer TL. To PLEX or not to PLEX in natalizumab-associated PML. Neurology. 2017;88(12):1108-9. Epub 20170222. doi: 10.1212/WNL.0000000000003747. PubMed PMID: 28228561.
- Tan CS, Bord E, Broge TA, Jr., Glotzbecker B, Mills H, Gheuens S, Rosenblatt J, Avigan D, Koralnik IJ. Increased program cell death-1 expression on T lymphocytes of patients with progressive multifocal leukoencephalopathy. J Acquir Immune Defic Syndr. 2012;60(3):244-8. doi: 10.1097/QAI.0b013e31825a313c. PubMed PMID: 22549384; PMCID: PMC3400136.
- Ercan MB, Kocer B, Altiparmak T, Arslan I. Responsiveness to pembrolizumab in severe early-onset natalizumab associated PML-IRIS in patient with relapsing-remitting multiple sclerosis. Mult Scler. 2024;30(6):760-2. Epub 20240222. doi: 10.1177/13524585241231655. PubMed PMID: 38385206.
- Jeantin L, Shor N, Pallix-Guyot M, Roos-Weil D, Bellanger A, Le Garff-Tavernier M, Papeix C, Weiss N, Pourcher V. Halting progressive multifocal leukoencephalopathy with pembrolizumab: the case of a patient with multiple sclerosis under fingolimod. J Neurol. 2024;271(2):729-32. Epub 20231101. doi: 10.1007/s00415-023-12055-9. PubMed PMID: 37910249.
- Cortese I, Muranski P, Enose-Akahata Y, Ha SK, Smith B, Monaco M, Ryschkewitsch C, Major EO, Ohayon J, Schindler MK, Beck E, Reoma LB, Jacobson S, Reich DS, Nath A. Pembrolizumab Treatment for Progressive Multifocal Leukoencephalopathy. N Engl J Med. 2019;380(17):1597-605. Epub 20190410. doi: 10.1056/NEJMoa1815039. PubMed PMID: 30969503.
- Roos-Weil D, Weiss N, Guihot A, Uzunov M, Bellanger A, Eymard B, Saadoun D, Houillier C, Idbaih A, Demeret S, Deback C, Leblond V, Galanaud D, Shor N, Pourcher V. Immune checkpoint inhibitors for progressive multifocal leukoencephalopathy: a new gold standard? J Neurol. 2021;268(7):2458-65. Epub 20210130. doi: 10.1007/s00415-021-10414-y. PubMed PMID: 33515299.
- Darcy S, Alexander M, McCarthy A, O'Dowd S. Pembrolizumab treatment of inflammatory progressive multifocal leukoencephalopathy: a report of two cases. J Neurovirol. 2022;28(1):145-50. Epub 20211207. doi: 10.1007/s13365-021-01028-1. PubMed PMID: 34874539.
- Boesl F, Allers K, Herm J, Scheider T, Franke C. Sequential treatment of progressive multifocal leukoencephalopathy with intravenous immunoglobulins and pembrolizumab. J Neurovirol. 2022;28(2):335-8. Epub 20220323. doi: 10.1007/s13365-022-01059-2. PubMed PMID: 35320511; PMCID: PMC9187551.
- Thaker AA, Schmitt SE, Pollard JR, Dubroff JG. Natalizumab-induced progressive multifocal leukoencephalopathy. Clin Nucl Med. 2014;39(7):e365-6. doi: 10.1097/RLU.0000000000000238. PubMed PMID: 24152618.
- Calic Z, Cappelen-Smith C, Hodgkinson SJ, McDougall A, Cuganesan R, Brew BJ. Treatment of progressive multifocal leukoencephalopathy-immune reconstitution inflammatory syndrome with intravenous immunoglobulin in a patient with multiple sclerosis treated with fingolimod after discontinuation of natalizumab. J Clin Neurosci. 2015;22(3):598-600. Epub 20141215. doi: 10.1016/j.jocn.2014.08.016. PubMed PMID: 25523125.
- Jamilloux Y, Kerever S, Ferry T, Broussolle C, Honnorat J, Seve P. Treatment of Progressive Multifocal Leukoencephalopathy With Mirtazapine. Clin Drug Investig. 2016;36(10):783-9. doi: 10.1007/s40261-016-0433-8. PubMed PMID: 27401779.
- Aoki H, Matsumura R, Nishida Y, Nakamichi K, Yokota T, Sanjo N. Efficacy of filgrastim therapy for patients with progressive multifocal leukoencephalopathy: Two case reports. Int J Infect Dis. 2025;158:107950. Epub 20250616. doi: 10.1016/j.ijid.2025.107950. PubMed PMID: 40532968.
- Lombardo-Del Toro P, Bragado-Trigo I, Arroyo P, Tena-Cucala R, Bau L, Matas E, Munoz-Vendrell A, Simo M, Pons-Escoda A, Martinez-Yelamos A, Martinez-Yelamos S, Romero-Pinel L. Fingolimod-associated progressive multifocal leukoencephalopathy in a multiple sclerosis patient with a good response to filgrastim. J Neurol. 2023;270(11):5196-200. Epub 20230717. doi: 10.1007/s00415-023-11865-1. PubMed PMID: 37460853.
- Stefoski D, Balabanov R, Waheed R, Ko M, Koralnik IJ, Sierra Morales F. Treatment of natalizumab-associated PML with filgrastim. Ann Clin Transl Neurol. 2019;6(5):923-31. Epub 20190408. doi: 10.1002/acn3.776. PubMed PMID: 31139690; PMCID: PMC6529830.
- Tan K, Roda R, Ostrow L, McArthur J, Nath A. PML-IRIS in patients with HIV infection: clinical manifestations and treatment with steroids. Neurology. 2009;72(17):1458-64. Epub 20090107. doi: 10.1212/01.wnl.0000343510.08643.74. PubMed PMID: 19129505; PMCID: PMC2677476.
- Clifford DB. Progressive multifocal leukoencephalopathy therapy. J Neurovirol. 2015;21(6):632-6. Epub 20140917. doi: 10.1007/s13365-014-0289-8. PubMed PMID: 25227934; PMCID: PMC4363307.
- Giacomini PS, Rozenberg A, Metz I, Araujo D, Arbour N, Bar-Or A, Maraviroc in Multiple Sclerosis-Associated PMLIG. Maraviroc and JC virus-associated immune reconstitution inflammatory syndrome. N Engl J Med. 2014;370(5):486-8. doi: 10.1056/NEJMc1304828. PubMed PMID: 24476450; PMCID: PMC5052063.
- Scarpazza C, Prosperini L, Mancinelli CR, De Rossi N, Lugaresi A, Capobianco M, Moiola L, Naldi P, Imberti L, Gerevini S, Capra R. Corrigendum to "Is maraviroc useful in multiple sclerosis patients with natalizumab-related progressive multifocal leukoencephalopathy?" [J. Neurol. Sci. 378 (2017) 233-237]. J Neurol Sci. 2017;380:19. Epub 20170704. doi: 10.1016/j.jns.2017.06.037. PubMed PMID: 28870565.
- Hodecker SC, Sturner KH, Becker V, Elias-Hamp B, Holst B, Friese MA, Heesen C. Maraviroc as possible treatment for PML-IRIS in natalizumab-treated patients with MS. Neurol Neuroimmunol Neuroinflamm. 2017;4(2):e325. Epub 20170208. doi: 10.1212/NXI.0000000000000325. PubMed PMID: 28210661; PMCID: PMC5299629.
- Martin-Blondel G, Cuzin L, Delobel P, Cuvinciuc V, Dumas H, Alvarez M, Massip P, Marchou B. Is maraviroc beneficial in paradoxical progressive multifocal leukoencephalopathy-immune reconstitution inflammatory syndrome management? AIDS. 2009;23(18):2545-6. doi: 10.1097/QAD.0b013e32833365f4. PubMed PMID: 19907215.
- Dong-Si T, Richman S, Wattjes MP, Wenten M, Gheuens S, Philip J, Datta S, McIninch J, Bozic C, Bloomgren G, Richert N. Outcome and survival of asymptomatic PML in natalizumab-treated MS patients. Ann Clin Transl Neurol. 2014;1(10):755-64. Epub 20141009. doi: 10.1002/acn3.114. PubMed PMID: 25493267; PMCID: PMC4241803.
- Dong-Si T, Gheuens S, Gangadharan A, Wenten M, Philip J, McIninch J, Datta S, Richert N, Bozic C, Bloomgren G, Richman S, Weber T, Clifford DB. Predictors of survival and functional outcomes in natalizumab-associated progressive multifocal leukoencephalopathy. J Neurovirol. 2015;21(6):637-44. Epub 20150314. doi: 10.1007/s13365-015-0316-4. PubMed PMID: 25771865; PMCID: PMC4628054.
- Crossley KM, Agnihotri S, Chaganti J, Rodriguez ML, McNally LP, Venna N, Turbett SE, Gutman M, Morey A, Koralnik IJ, Brew BJ. Recurrence of progressive multifocal leukoencephalopathy despite immune recovery in two HIV seropositive individuals. J Neurovirol. 2016;22(4):541-5. Epub 20160104. doi: 10.1007/s13365-015-0419-y. PubMed PMID: 26727910; PMCID: PMC4925684.
- Sanchez-Quintana A, Brena-Atienza J, Marrero-Santos C, Alvarez-Acosta L. Late relapse of progressive multifocal leucoencephalopathy postallogenic transplant in a young patient with CLL. BMJ Case Rep. 2013;2013. Epub 20130805. doi: 10.1136/bcr-2013-200213. PubMed PMID: 23917368; PMCID: PMC3762402.
- Maillart E, Vidal JS, Brassat D, Stankoff B, Fromont A, de Seze J, Taithe F, Clavelou P, Bourre B, Delvaux V, Rico A, Labauge P, Tourbah A, Lebrun C, Pelletier J, Moreau T, Louapre C, Lubetzki C, Papeix C. Natalizumab-PML survivors with subsequent MS treatment: Clinico-radiologic outcome. Neurol Neuroimmunol Neuroinflamm. 2017;4(3):e346. Epub 20170414. doi: 10.1212/NXI.0000000000000346. PubMed PMID: 28616447; PMCID: PMC5462603.
- O'Hara BA, Gee GV, Haley SA, Morris-Love J, Nyblade C, Nieves C, Hanson BA, Dang X, Turner TJ, Chavin JM, Lublin A, Koralnik IJ, Atwood WJ. Teriflunomide Inhibits JCPyV Infection and Spread in Glial Cells and Choroid Plexus Epithelial Cells. Int J Mol Sci. 2021;22(18). Epub 20210910. doi: 10.3390/ijms22189809. PubMed PMID: 34575975; PMCID: PMC8468119.