Hypoplastic left heart syndrome (HLHS) is a heart condition that develops before birth (congenital). The left side of a baby’s heart doesn’t form right. This keeps blood from flowing through their heart in a normal way. Medicine can help shortly after birth, but babies with HLHS need a series of three surgeries at very young ages.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
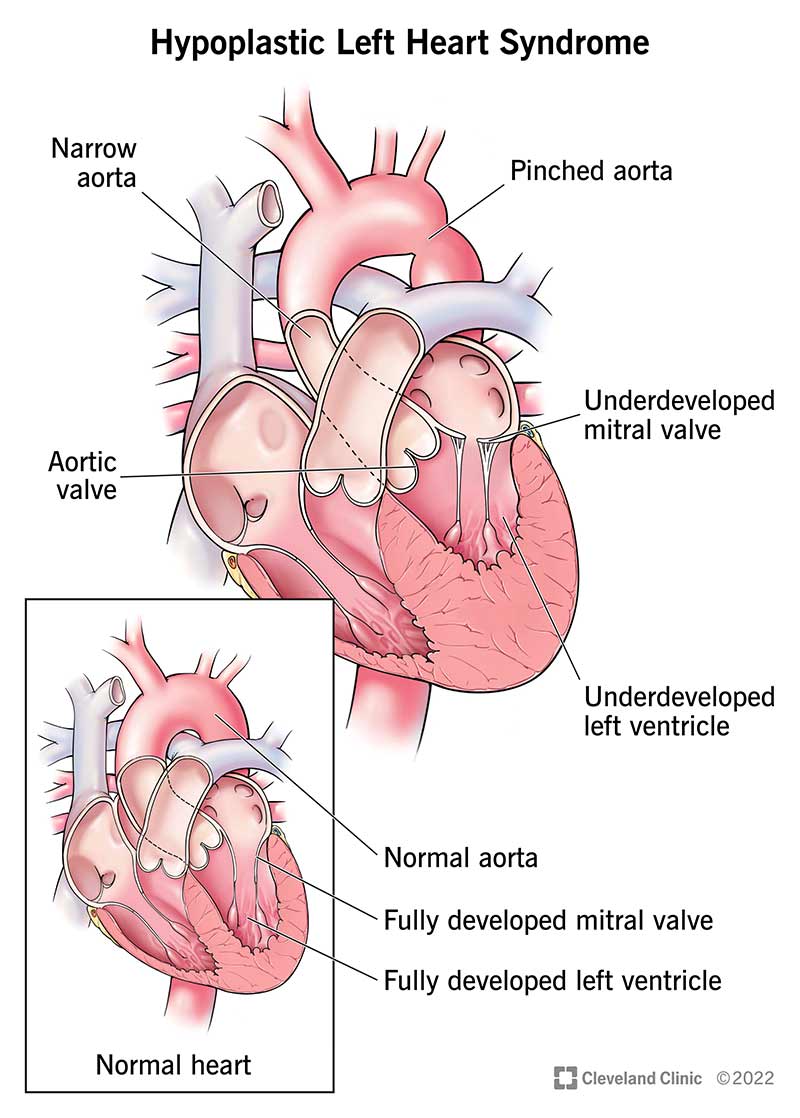
Image content: This image is available to view online.
View image online (https://my.clevelandclinic.org/-/scassets/images/org/health/articles/12214-hypoplastic-left-heart-syndrome)
Hypoplastic left heart syndrome (HLHS) is a complex and rare heart condition that’s present at birth (congenital). It’s a single-ventricle heart defect. This means one side of your baby’s heart didn’t develop correctly.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
In babies born with hypoplastic left heart syndrome, some structures in the left side of their heart didn’t develop completely, including the:
A baby with hypoplastic left heart syndrome may also have an opening (atrial septal defect) between the two upper heart chambers. Normally, this would be a solid wall of tissue.
Each side of a normal heart has a job. The right side moves oxygen-poor blood to your lungs to get oxygen. The left side sends oxygen-rich blood to your body.
In a baby with hypoplastic left heart syndrome, the heart’s left side is too small to pump enough blood to the rest of the body. Their right ventricle or right lower chamber of their heart then takes over to pump blood to both their lungs and whole body. It does this through a blood vessel called the ductus arteriosus.
Advertisement
The ductus arteriosus is a vessel that all fetuses have. It typically gets smaller until it closes soon after birth. With a closed ductus arteriosus, hypoplastic left heart syndrome is usually fatal if babies don’t receive treatment.
About 1 in 3,800 babies are born with hypoplastic left heart syndrome each year in the U.S. This condition accounts for about 2% to 3% of all congenital heart diseases (CHD). Hypoplastic left heart syndrome affects more male babies than female babies.
Hypoplastic left heart syndrome symptoms may include:
Newborns with HLHS don’t always show symptoms immediately. Instead, symptoms may develop within a few hours or days of birth.
Most of the time, there is no known cause for hypoplastic left heart syndrome. Sometimes, the cause is genetic. Babies with mutations (changes) in specific genes may have a higher risk of HLHS. These genes are known as GJA1 or NKX2-5.
Some babies with genetic disorders (such as Turner syndrome or trisomy 18) may also have hypoplastic left heart syndrome.
A healthcare provider can make a hypoplastic left heart syndrome diagnosis with information from harmless imaging.
Your provider may do this during your pregnancy or soon after your baby is born.
During pregnancy, healthcare providers use painless and non-invasive imaging methods such as:
After your baby is born, healthcare providers diagnose the condition by observing symptoms and examining test results. They may hear a heart murmur while using a stethoscope to listen to your baby’s heart. This means blood isn’t flowing the way it should.
Tests for a hypoplastic left heart syndrome diagnosis may include:
Yes. First, your baby will need a medication called a prostaglandin. This keeps their ductus arteriosus open and functioning. Other medicines can help your baby’s heart work more efficiently. Your baby also may need help with breathing.
Advertisement
Then they’ll need a series of surgeries to direct blood flow to their lungs and body. These operations put their heart’s workload on their right ventricle, which does all the pumping.
Surgeons perform three separate operations: the Norwood, Glenn and Fontan procedures. Your baby’s provider will need to do the surgeries in that order.
Norwood procedure: Babies with HLHS need Norwood surgery within the first two weeks of life. During the procedure, surgeons:
Bidirectional Glenn shunt operation: At 4 to 6 months of age, babies need a second operation. During the Glenn procedure, surgeons:
Advertisement
Fontan procedure: Between 18 months and 4 years of age, babies need a final surgery. This procedure allows all blood returning from their body to go straight to their lungs instead of mixing in their heart. During a Fontan procedure, surgeons will connect your baby’s inferior vena cava (IVC) to their pulmonary arteries. Similar to the SVC, the IVC is the large vein that carries oxygen-poor blood from your child’s lower body to their heart.
Some babies may not make it from one surgery to the next. Also, they may have problems after each surgery, such as:
Sometimes, your baby’s surgeon may recommend a heart transplant instead of the three-part surgery series. However, babies with heart transplants need lifelong medications.
Operating during pregnancy is not an option for treating hypoplastic left heart syndrome. A fetal surgeon may choose to operate only to correct some conditions that might be associated with poor HLHS outcomes in babies who are developing HLHS.
After treatment for hypoplastic left heart syndrome, your child will need lifelong follow-up visits at least once a year with a cardiologist (heart doctor). These visits ensure that their heart, lungs and other organs continue to work properly. When your child becomes an adult, they’ll transition to care with a specialist in adult congenital heart disease.
Advertisement
Most children with hypoplastic left heart syndrome will need heart medications. They’ll also need to take antibiotics before any other surgeries, including dental surgeries. These medications reduce the risk of endocarditis (a heart infection).
Without treatment, hypoplastic left heart syndrome is fatal days or weeks after birth.
With treatment, the prognosis depends on the complexity of your child’s heart deformity. Ask your baby’s healthcare provider about the risks associated with each surgery.
Some children may have decreased physical endurance for the rest of their lives. Usually, healthcare providers recommend limiting intensive physical activities, such as competitive sports.
About 20% to 60% of babies with hypoplastic left heart syndrome survive their first year of life. After that, the survival rate for the next five, 10 and 15 years is about 40%. Babies who have a normal birth weight and aren’t born prematurely do better than babies with lower birth weights. One study found that most babies who survived their first year were still alive at age 18.
Many HLHS cases don’t have an obvious cause. However, providers always encourage healthful habits during pregnancy, including:
If you or your partner has hypoplastic left heart syndrome in their family, you may want to talk with a genetic counselor before getting pregnant.
Children born with this condition can live a healthy life with long-term monitoring from a cardiologist.
You can care for your child in these ways:
If your baby has hypoplastic left heart syndrome, you may want to ask your healthcare provider:
Having a child with hypoplastic left heart syndrome can be stressful and lonely. It’s important to find a support group or other source of emotional support while you deal with the stresses and uncertainties of your child’s heart defect. Feeling emotionally strong will help you manage the highs and lows of your child’s health.

Sign up for our Health Essentials emails for expert guidance on nutrition, fitness, sleep, skin care and more.
Learn more about the Health Library and our editorial process.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
If your child has hypoplastic left heart syndrome, you want expert, compassionate help. At Cleveland Clinic Children’s, we offer the best care for your family.
