Rheumatoid arthritis is a type of arthritis where your immune system attacks the tissue lining the joints on both sides of your body. It may affect other parts of your body, too. The exact cause is unknown. Treatment options include lifestyle changes, physical therapy, occupational therapy, nutritional therapy, medication and surgery.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
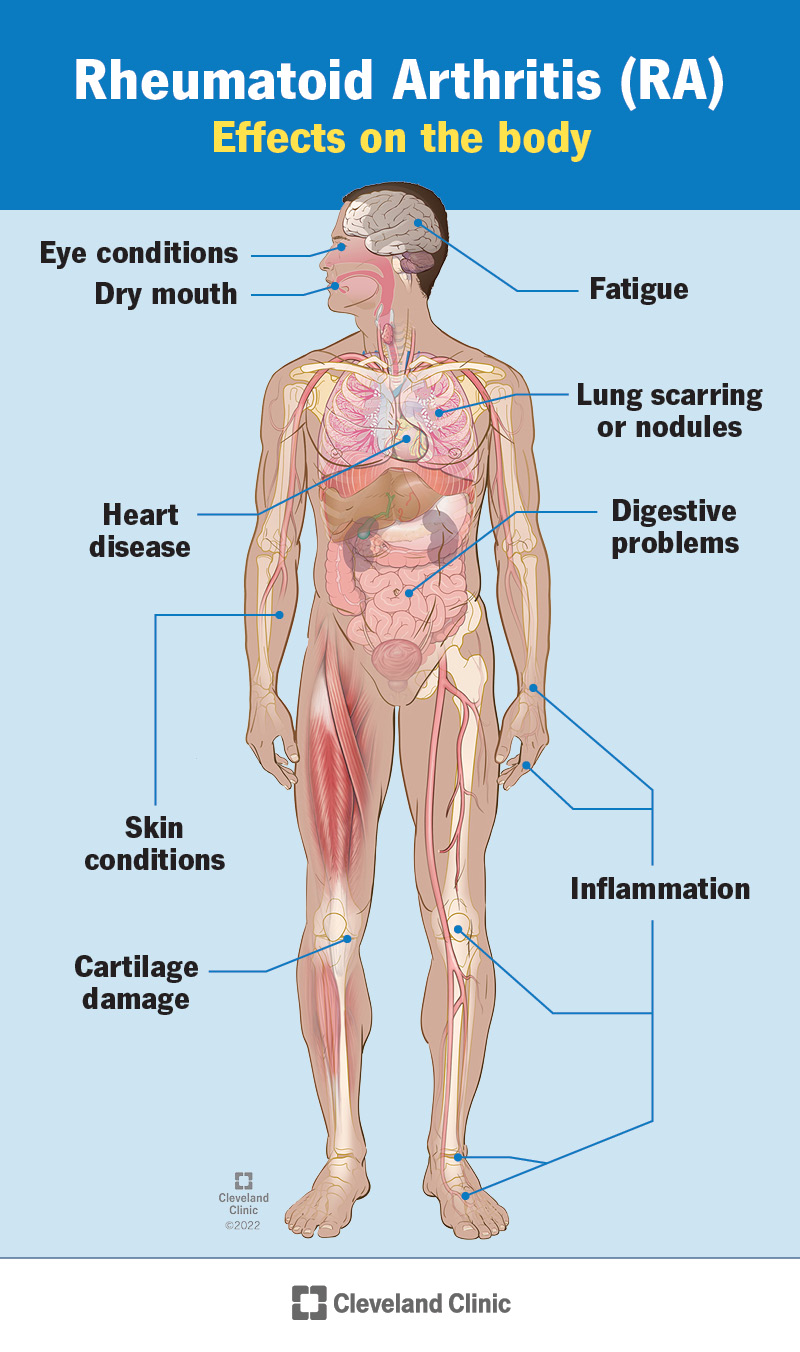
Image content: This image is available to view online.
View image online (https://my.clevelandclinic.org/-/scassets/images/org/health/articles/4924-rheumatoid-arthritis-2)
Rheumatoid arthritis (RA) is a chronic (ongoing) autoimmune disease that causes pain, swelling and stiffness in the lining of your joints (synovium). It most commonly affects the joints in your fingers, hands, wrists, knees, ankles, feet and toes. RA usually occurs in the same joints on both sides of your body, which makes it different from some other types of arthritis.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Uncontrolled inflammation damages cartilage, which normally acts as a “shock absorber” in your joints. In time, this can deform your joints. Eventually, your bone itself erodes. This can lead to the fusion of your joint(s).
Specific cells in your immune system (your body’s infection-fighting system) aid this process. These substances are produced in your joints but also circulate and cause symptoms throughout your body. In addition to affecting your joints, rheumatoid arthritis sometimes affects other parts of your body, including your:
The four stages of rheumatoid arthritis are:
Advertisement
It may take many years to go through all stages, and some people don’t progress through all stages.
RA usually starts to develop between the ages of 30 and 60. But anyone can develop the condition. In children and young adults — usually between the ages of 16 and 40 — it’s called young-onset rheumatoid arthritis (YORA). In people who develop symptoms after they turn 60, it’s called later-onset rheumatoid arthritis (LORA).
Symptoms of rheumatoid arthritis may include:
RA affects everyone differently. In some people, symptoms develop over several years. In other people, rheumatoid arthritis symptoms progress rapidly. Many people have time with symptoms (flares) and then time without any symptoms (remission).
Early signs of rheumatoid arthritis include tenderness or pain in small joints like those in your fingers or toes. Or you might notice pain in a larger joint like your knee or shoulder. These early signs of RA are like an alarm clock set to vibrate. It might not always be enough to get your attention. But the early signs are important because the sooner you’re diagnosed with RA, the sooner your treatment can begin. Also, prompt treatment lowers your risk of developing permanent, painful joint damage.
The exact cause of rheumatoid arthritis is unknown. Researchers think it’s caused by a combination of genetics, hormones and environmental factors.
Normally, your immune system protects your body from disease. With RA, something triggers your immune system to attack your own joints. An infection, smoking, or physical or emotional stress may be triggers.
Scientists have studied many genes as potential risk factors for RA. Certain genetic variations and nongenetic factors contribute to your risk of developing rheumatoid arthritis. Nongenetic factors include sex and exposure to irritants and pollutants.
People born with variations in the human leukocyte antigen (HLA) genes are more likely to develop rheumatoid arthritis. HLA genes help your immune system tell the difference between proteins your body makes and proteins from invaders like viruses and bacteria.
There are several risk factors for developing rheumatoid arthritis. These include:
Advertisement
Without treatment, rheumatoid arthritis can lead to further complications, including:
To make a rheumatoid arthritis diagnosis, your healthcare provider may refer you to a doctor who specializes in arthritis (rheumatologist). Rheumatologists diagnose people with RA based on a combination of several factors. They’ll do a physical exam and ask you about your medical history and symptoms. Your rheumatologist will also order blood tests and imaging tests.
The blood tests look for inflammation and blood proteins (antibodies) that are signs of rheumatoid arthritis. These may include:
Your rheumatologist may order imaging tests to look for signs that your joints are wearing away. Rheumatoid arthritis can cause the ends of the bones within your joints to wear down. The imaging tests may include:
In some cases, your provider may watch how you do over time before making a definitive diagnosis of RA.
Advertisement
The most important goal of treating rheumatoid arthritis is to reduce joint pain and swelling. Doing so should help maintain or improve joint function. The long-term goal of treatment is to slow or stop joint damage. Managing joint inflammation reduces your pain and improves your quality of life.
Rheumatoid arthritis treatments typically include one or more kinds of medication. In certain cases, your provider may recommend surgery. Your provider considers your age, health, medical history and how bad your symptoms are when deciding on a treatment.
Early treatment with certain drugs can improve your long-term outcome. A combination of drugs may be more effective than, and appear to be as safe as, single-drug therapy.
There are many medications to decrease joint pain, swelling and inflammation, as well as to prevent or slow down the disease. Medications that treat rheumatoid arthritis include:
The safest drug for rheumatoid arthritis is one that gives you the most benefit with the least number of side effects. This varies depending on your health history and the severity of your RA symptoms. Your healthcare provider will work with you to develop a treatment program.
Advertisement
Surgery may be an option to restore function to severely damaged joints. Your provider may also recommend surgery if your pain isn’t managed with medication. Surgeries that treat RA include:
Although there isn’t currently a cure for rheumatoid arthritis, there are many effective methods for decreasing your pain and inflammation and slowing down the disease process. Early diagnosis and effective treatment are very important.
If you don’t see a provider for RA treatment, the disease can cause permanent damage to your cartilage and, eventually, your joints. RA can also harm organs like your lungs and heart.
It’s important to see your healthcare provider on a regular basis to monitor your symptoms. They’ll also want to know about any side effects you may experience from your medications. Your provider can adjust your dosage or change the types of medications you take. Continue to take your medications until you speak with your provider.
You can also take care of yourself by following a healthy eating plan and getting some physical activity every day. If you smoke, it’s important that you quit.
Questions you may want to ask your provider include:
If you have rheumatoid arthritis (RA), you may feel like you’re on a lifelong roller coaster of pain and fatigue. It’s important to share these feelings and your symptoms with your healthcare provider. Along with X-rays and blood tests, what you say about your quality of life will help inform your treatment. Your healthcare provider will assess your symptoms and recommend the right treatment plan for your needs. Most people can manage rheumatoid arthritis and still do the activities they care about.

Sign up for our Health Essentials emails for expert guidance on nutrition, fitness, sleep, skin care and more.
Learn more about the Health Library and our editorial process.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
Rheumatoid arthritis is a chronic condition, but with the right treatment, you can manage it. Cleveland Clinic is here to help you live an active life.
