Chimeric antigen receptor (CAR) T-cell therapy treats some types of blood cancer. Scientists create the treatment by adding a lab-made gene to your cancer-fighting T cells. The change helps T cells detect and kill cancerous cells. Healthcare providers may use this treatment when other treatments aren’t effective or blood cancer comes back.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
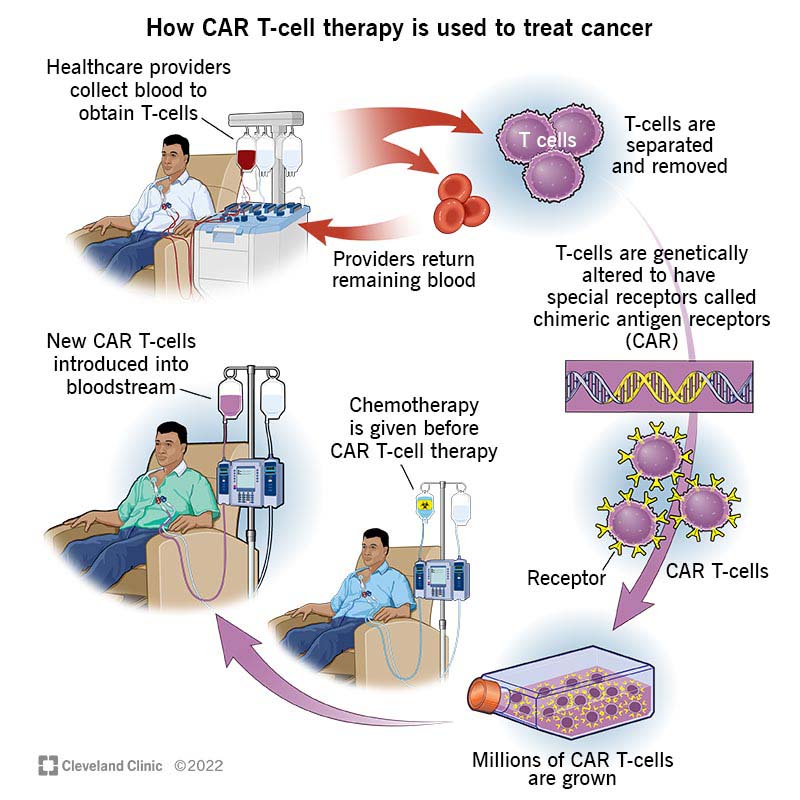
Image content: This image is available to view online.
View image online (https://my.clevelandclinic.org/-/scassets/Images/org/health/articles/17726-car-t-cell-therapy)
Chimeric antigen receptor (CAR) T-cell therapy is a type of immunotherapy for some types of blood cancer. It works by turning your T lymphocytes (T cells) into more efficient cancer-fighting machines. In CAR T-cell therapy, healthcare providers introduce a new gene into your T cells that changes your cells so they can do more to detect and kill cancerous cells. In some cases, CAR T-cell therapy can cure blood cancer. Other times, it helps people with certain blood cancers live longer.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Right now, CAR T-cell therapy is an option to treat some blood cancers when other treatments aren’t effective or the condition comes back. Those blood cancer types include:
To understand how CAR T-cell therapy works, it may help to know more about T cells, which are white blood cells in your immune system. Your immune system monitors your body for intruders, including cancer, by tracking proteins called antigens on the surface of intruder cells.
Your T cells have their own surface proteins called receptors. These receptors can recognize cells that have abnormal antigens. T cells act as a surveillance system for abnormal cells, becoming active when a receptor recognizes an abnormal cell. The activated T cell goes to work, destroying the abnormal cell and activating other parts of your immune system to come help find and destroy abnormal cells.
But your T-cell receptors don’t always detect cancerous cells. Enter CAR T cells, your own T cells that are changed so they recognize a specific antigen on the surface of cancer cells. Scientists change your T cells by adding a lab-made gene for a chimeric antigen receptor. Next, they make the new CAR T cells grow and multiply until there are enough cells to target cancerous cells effectively.
Advertisement
Once in your bloodstream, CAR T-cell receptors detect and destroy cancerous cells. The cells also keep on multiplying so you have a long-term supply of CAR T cells targeting your cancer cells. That long-term supply is why researchers and healthcare providers call CAR T-cell therapy a type of “living drug.”
Yes, there are. Right now, there are six CAR T-cell therapies with U.S. Food and Drug Administration (FDA) approval to treat specific kinds of blood cancer. Those therapies are:
First, you donate white blood cells through a procedure called leukapheresis. In leukapheresis, a machine draws some of your blood, extracts your white blood cells and then returns the remaining blood to your body. Typically, your providers will insert a central venous catheter to remove and return your blood.
Next, your white blood cells are quickly transported to the facility where scientists will turn your cells into CAR T cells. This process can take between three and six weeks.
Depending on your situation, you may have chemotherapy or radiation therapy after leukapheresis but before you receive CAR T cells. Your healthcare provider may call this bridging therapy. These treatments manage blood cancer during the time it takes to produce and grow enough CAR T cells to treat your condition.
When it’s time for you to receive your new cells, you’ll undergo lymphodepleting chemotherapy. This treatment keeps your body from rejecting the CAR T cells.
The actual treatment involves receiving CAR T cells via infusion. CAR T-cell infusion takes between five and 30 minutes and is done via an intravenous line (IV). You may need to stay in the hospital during the procedure. The infusion process is like a blood transfusion:
This treatment may cause serious side effects, so your provider may recommend that you stay in the hospital immediately after treatment. Depending on your situation, including side effects, you may need to stay in the hospital for seven to 10 days. After that, you should plan to be within driving distance of your treatment center for the first month after treatment. You’ll also need:
Advertisement
The two most common side effects are cytokine release syndrome and neurological issues. Cytokine release syndrome is an inflammatory syndrome that causes symptoms, including:
Neurological issues from CAR T-cell therapy may include:
Other potential side effects are:
CAR T-cell therapy is a newer way to treat some kinds of blood cancer when other treatments aren’t effective or when cancer comes back (recurs). Sometimes, CAR T-cell therapy can cure certain types of blood cancer. Often, it helps people live longer.
A 2020 study tracked children receiving treatment for acute lymphoblastic leukemia. More than 85% of the children had complete remission right after CAR T-cell treatment and 60% of those children remained free of cancer 12 months after treatment. On the other hand, an analysis of CAR T-cell therapy research concluded that for a significant percentage of people, CAR T-cell therapy wasn’t a cure for their condition.
Advertisement
CAR T-cell therapy side effects can be life-threatening, especially cytokine release syndrome and some neurological issues. Before you leave the hospital, be sure to ask your provider about symptoms that could be signs of serious issues. For example, cytokine release syndrome symptoms that linger, like fever, chills, headache and nausea, may be reason to call your provider right away or go to the emergency room.
CAR T-cell therapy continues to bring hope for many people with blood cancer. For some, CAR T-cell therapy represents the hope of living longer when other treatments aren’t effective or blood cancer comes back. For others, the treatment brings hope for a cure. This treatment is rapidly changing how healthcare providers treat cancer.
Since 2017, the FDA has approved CAR T-cell therapies. Researchers are overseeing more than 1,000 clinical trials to test potential CAR T-cell drugs. If you have blood cancer, ask your healthcare provider if this treatment is an option. If you have another kind of cancer, ask your providers about participating in a clinical trial.
Advertisement

Sign up for our Health Essentials emails for expert guidance on nutrition, fitness, sleep, skin care and more.
Learn more about the Health Library and our editorial process.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
When you’re diagnosed with cancer, you want expert and compassionate care right away. At Cleveland Clinic we personalize your treatment to match your needs.
