Fecal transplantation means transferring a medically processed fecal sample from one person’s gut to another. The thousands of microorganisms contained within the sample have therapeutic benefits. Most of these benefits are still in the research phase. However, healthcare providers do prescribe fecal transplants for recurrent C. diff infections.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
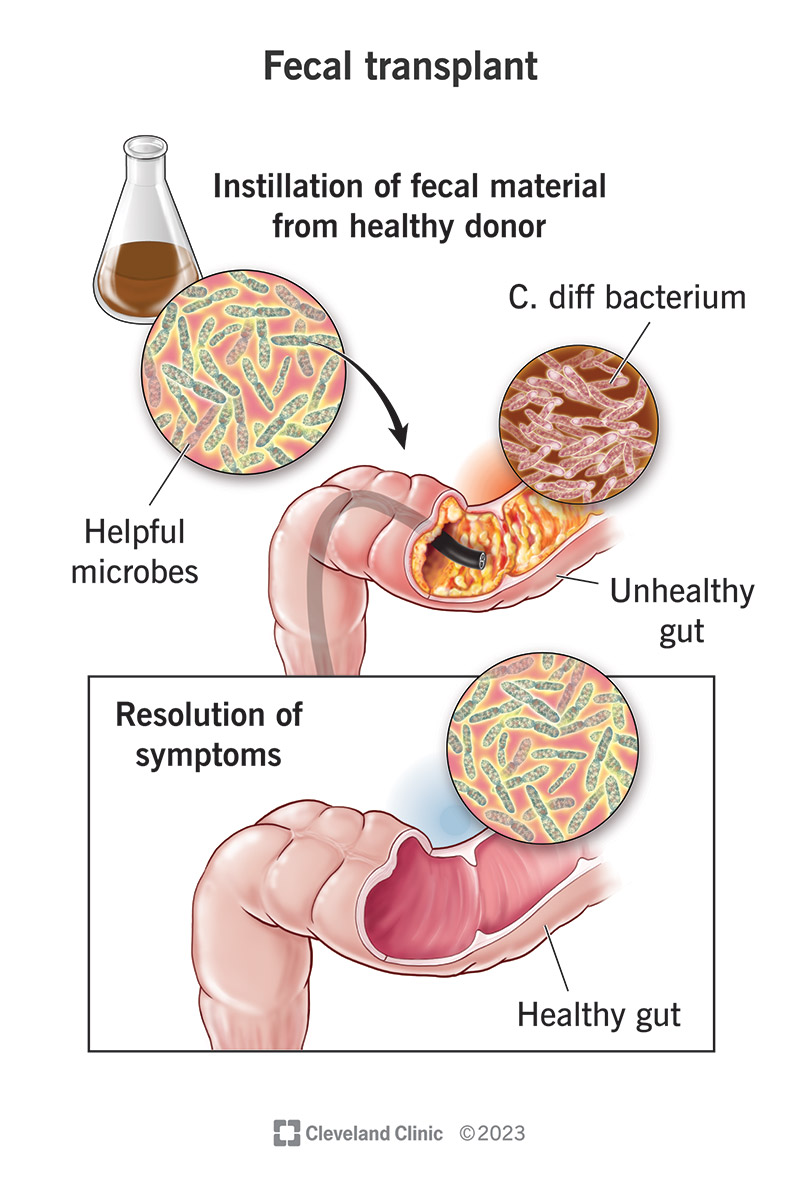
Image content: This image is available to view online.
View image online (https://my.clevelandclinic.org/-/scassets/Images/org/health/articles/25202-fecal-transplant)
Fecal microbiota transplantation (FMT) is a medical procedure to transplant a small sample of stool (feces) from a healthy colon into a diseased colon. Each healthy stool sample contains thousands of beneficial microbiota that can improve the health of the diseased colon in a variety of ways.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Healthcare providers currently prescribe fecal transplantation to treat a severe bacterial infection in your colon. The transplant transfers a sampling of helpful microbiota into the infected colon to help fight off the pathogenic ones. This helpful microbiota can restore balance to your gut microbiome.
Every person’s gut is home to trillions of microorganisms, most of which are beneficial. These helpful microbes perform many important services in your body, including preventing unhelpful germs from taking over. But drugs, diseases and other factors can harm and reduce your supply of helpful microbes.
Your gut microbiome is a delicate ecosystem. When unhelpful microbes outnumber the helpful ones, that balance gets disrupted. A fecal transplant is a simple and effective way to restore a healthy microbial mix. The thousands of helpful microbes in a healthy stool sample help repopulate your gut.
Video content: This video is available to watch online.
View video online (https://cdnapisec.kaltura.com/p/2207941/sp/220794100/playManifest/entryId/1_atiqtynd/flavorId/1_5f3sgelj/format/url/protocol/https/a.mp4)
Learn about fecal transplants from Gail Cresci, PhD, RD.
Fecal transplant is currently recommended and proven effective only as a treatment for persistent C. difficile infection (CDI). C. diff is an invasive bacterium that can be resistant to treatment with antibiotics. When antibiotics fall short, fecal transplantation is often successful in treating C. diff infection.
Advertisement
However, fecal transplantation may be beneficial for many other diseases. Your gut microbiome has wide-ranging effects on your digestive system, your metabolism and your immune system. Healthcare providers are researching fecal transplant as a possible treatment for many conditions, including:
Fecal microbiota transplantation is still a new and investigational treatment for C. diff infection. Most healthcare providers only prescribe it after other treatments have repeatedly failed. However, microbiota restoration therapies (MRTs) are generally considered the future of C. diff treatment.
Fecal microbiota can be delivered to your GI tract in a variety of ways. Historically, healthcare providers have used different methods in different circumstances. Currently, only two fecal microbiota therapies have been approved by the U.S. Food and Drug Administration (FDA): an enema and an oral capsule.
Fecal transplant methods include:
Advertisement
There’s no preparation required for the fecal microbiota, live-jslm enema (RBL, Rebyota). You don’t need to prepare your bowels with laxatives as you would before a colonoscopy, and you won’t need anesthesia for the procedure. This makes it accessible to a wider range of people who may need it.
You’ll receive the transplant in one dose in your healthcare provider’s office, within one to three days after finishing antibiotic therapy for C. diff infection. It only takes a few minutes. For the procedure, you’ll put on a hospital gown and lie on a table on your left side with your knees bent to your chest.
The fecal transplant enema comes in a bag connected to a tube with a clamp. Your provider will apply a lubricant inside your anus to make the enema more comfortable. Then they’ll gently insert the tube (about 5 inches), release the clamp and raise the bag, allowing the contents to flow in by gravity.
When the delivery is complete, your provider will withdraw the tube. You’ll continue to lie in the same position for about 15 minutes. This helps to prevent cramping and leaking. Some providers may give you medication to slow your bowels, to make sure the transplant stays inside long enough to take hold.
Advertisement
You can begin taking SER-109 (fecal microbiota spores, live-brpk) oral capsules by prescription within two to four days of finishing your course of antibiotics. You’ll take the prescription home with you. You’ll have one day of prep before starting. Then you’ll swallow four pills a day for the next three days.
The prep involves fasting and using a laxative formula to clear out your bowels. The suggested laxative is a magnesium citrate solution, though this might not be appropriate for people with certain health conditions. Check with your healthcare provider first if you have diabetes or chronic kidney disease (CKD).
The laxative comes as a powder that you’ll mix with 10 ounces of water. You’ll drink this solution at least eight hours before taking your first dose of SER-109. You’ll usually follow it with at least one full glass of water. After that, you’ll stop eating and drinking for the eight hours before taking your first dose.
The laxative will produce one to several bowel movements during the next eight hours. By the following morning, your bowels should be clear and ready to receive your first dose of fecal microbiota. Each of the next three mornings, you’ll take your dose on an empty stomach, before resuming food and drink.
Advertisement
Volunteer fecal donors provide stool samples for fecal transplants. Healthcare providers rigorously screen volunteers before accepting them as fecal donors. They make sure the donor stool is free of any infections or diseases. After collecting the stool (about 50 grams), they send it to a lab for processing.
Fecal donations are blended with sterile saline and then filtered, producing a liquid solution. The liquid might be used right away, especially if it’s meant for someone in particular. If not, it goes to a donor bank. The sample might be frozen to be thawed later, or it might be freeze-dried and put into capsules.
Your fecal transplant donor may be someone you know, or they may be an anonymous donor who submits to a frozen donor bank. The two fecal microbiota products currently approved by the FDA are manufactured in advance. The manufacturers source, screen and process their own donor material.
Fecal donor volunteers take blood tests and stool tests that screen for various gastrointestinal diseases that might be transferred with their stool. They may also take a urinalysis and oral medical history. Volunteers may be ruled out as potential fecal donors if their evaluation reveals any of the following:
C. diff is a common gut bacterium that lives in many healthy colons without causing harm. Only when the helpful microbes in your gut are diminished does C. diff run rampant. Healthcare providers recognize that a healthy gut microbiome is an important factor in preventing and managing this disease.
Only a few antibiotics can treat C. diff infection, and even these don’t always work. When they do work, they only kill the bacteria themselves, but not the spores. As long as your gut microbiome remains weak, C. diff infection can return. In some people, it returns again and again, coming back stronger each time.
By seeding other types of microorganisms in your gut, fecal microbiota transplantation helps to reestablish a healthy gut microbiome that can stand up to C. diff. These more beneficial bacteria compete with C. diff spores, preventing the infection from returning after antibiotic treatment.
The FDA has approved two fecal microbiota products specifically to prevent recurrent C. diff infection. These products come with a disclaimer that they aren’t approved to treat C. diff. That’s just to ensure that they aren’t the only treatment. The best approach is to use them following antibiotic treatment.
Success rates vary between different delivery methods, but overall, FMT is 80% to 95% successful in preventing C. diff infection from recurring after antibiotic treatment. This represents an important breakthrough in the treatment of recurrent CDI, which has been a serious medical challenge.
You may experience mild to moderate gastrointestinal (GI) symptoms after the treatment, like bloating and gas, nausea and vomiting, or abdominal discomfort. Some people get a fever and chills. These are expected side effects and shouldn’t be cause for concern. They should pass within a day or two.
There’s a remote but serious risk that a fecal transplant could transfer a harmful pathogen to the recipient. Bacteria and viruses that were inactive or were well managed within the donor’s body may become active or spread out of control within the recipient’s body, due to their weakened condition.
Fecal transplant samples are rigorously screened and processed to mitigate this risk. But since there aren’t yet standardized protocols for the screening or processing, it’s impossible to guarantee the safety of all fecal transplants. This is why the FDA has only approved two select fecal microbiota products.
FDA approval means that these products have passed the necessary clinical trials and review process to be considered safe for therapeutic use. While the labels on these products admit that a theoretical risk of infection remains, the products haven’t been found to cause infections or other adverse effects.
Both products are produced using Good Manufacturing Practices (GMP). Manufacturers of the fecal transplant enema RBL conduct regular, repeat testing of their fecal samples and donors. Manufacturers of the fecal transplant pill SER-109 purify their samples, preserving only the (safe) Firmicutes spores.
The idea of poop as medicine is bound to produce a certain gut reaction. Maybe you find its natural, organic qualities appealing, or maybe you find it, well, stranger than fiction. But for those people facing another round of relentless C. diff diarrhea and colitis, this novel idea represents, above all, hope.
In fact, the concept of fecal microbiota transplantation in medicine dates back centuries. What’s more recent is our growing understanding of these microbiota. The more we learn, the more hope we have that, in time, they may offer answers not only to C. diff infection but to many other intractable diseases.

Sign up for our Health Essentials emails for expert guidance on nutrition, fitness, sleep, skin care and more.
Learn more about the Health Library and our editorial process.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
If you have issues with your digestive system, you need a team of experts you can trust. Our gastroenterology specialists at Cleveland Clinic can help.
