
Joe Schwappach, a fleet manager for a pork company, suffered an arm injury on the job while helping herd pigs who had escaped from an overturned truck. He expected the 45-minute surgery near his home, outside St. Louis, would be simple. He even planned to get ice cream afterwards.
Instead, the 50-year-old resident of Grainfort, Illinois, vaguely remembers being transported to an Omaha, Nebraska, hospital after suffering a stroke during the outpatient procedure to repair his torn bicep. It paralyzed most of his right side and left him with limited ability to speak. He spent four months in two Omaha rehabilitation facilities, undergoing physical, speech and occupational therapy.
Joe slowly regained his speech and ability to walk, both of which are now nearly back to normal. But he had little control of his dominant right hand, which he needed as part of his job managing a team of truck mechanics and often pitching in to help.
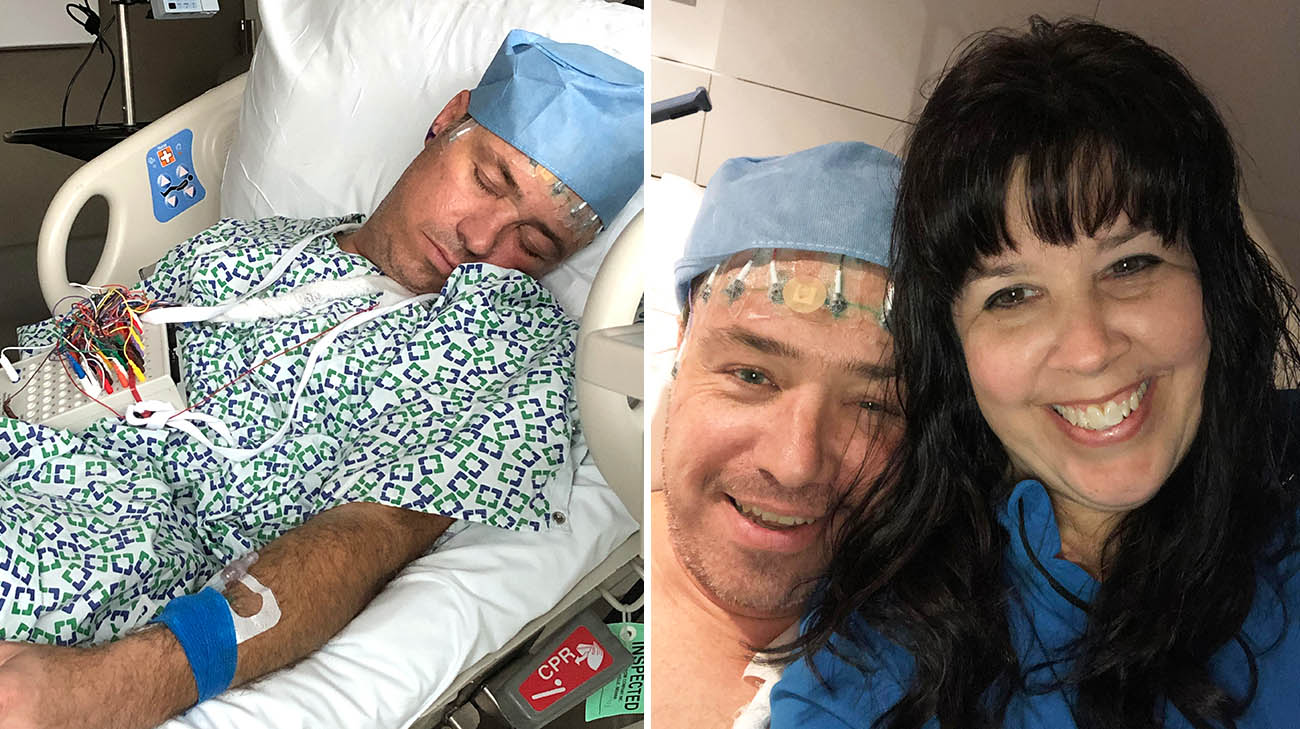
Joe with his wife, Stephanie, after his first DBS surgery. (Courtesy: Stephanie Schwappach)
“I’m naturally right-handed, and I couldn’t brush my teeth, tie my shoes or move a coffee cup up or down,” Joe recalls. “I had made a lot of progress, but my hand wasn’t getting much better.”
Upon returning home in June of 2018, he visited a neurologist who suggested Joe investigate whether there were any clinical trials for treatments. He and his wife, Stephanie, poured through information online.
They discovered Cleveland Clinic’s deep brain stimulation (DBS) study for patients with weakness after stroke, the number one cause of long-term disability in the U.S. After calling to seek more information, Joe found himself in Cleveland, Ohio, two weeks later for tests and exams. He was accepted into the EDEN trial (Electrical Stimulation of the Dentate Nucleus for Upper Extremity Hemiparesis Due to Ischemic Stroke), which is supported by the National Institutes of Health’s BRAIN initiative and Enspire DBS Therapy. It’s the first in-human trial of DBS for patients undergoing post-stroke rehabilitation. Joe is one of 12 participants in the trial.

Deep brain stimulation is a surgical procedure that involves implanting electrodes in the brain, which deliver electrical impulses that block or change the abnormal activity that cause symptoms. (Courtesy: Cleveland Clinic)
“He had limited use of his hand. It really wasn’t functioning,” explains Andre Machado, MD, PhD, who is Chairman of the Cleveland Clinic Neurological Institute. “He couldn’t do as much manual work, with both hands, and that was a limitation for his quality of life.”
For the study, Joe first underwent two surgical procedures -- one to insert the DBS device under the skin of his chest, just below the collarbone, and the other, to implant the DBS electrode in a part of the cerebellum called the dentate nucleus. Once activated, the device, called an implantable pulse generator, serves as a specially-calibrated pacemaker for the brain, stimulating it to try and enhance motor rehabilitation.
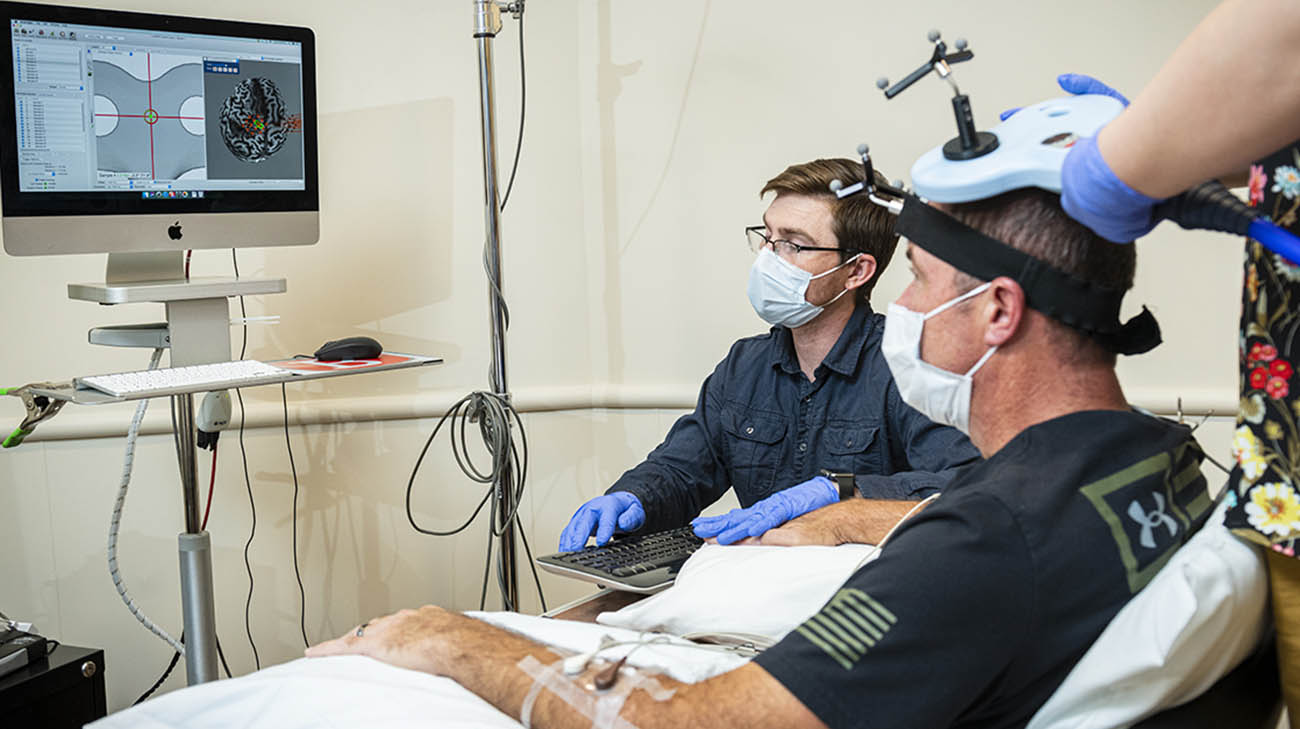
Joe underwent therapy and testing to help build his functionality. (Courtesy: Cleveland Clinic)
Joe said wearing the device came with no side effects. Initially, he came to Cleveland Clinic several times to have the device calibrated. Researchers reviewed his brain wave activity and effects of the stimulation. He would then come three times a week to undergo virtual physical therapy, with a Cleveland Clinic therapist, who would lead him through various activities. Joe would return monthly to Cleveland Clinic for follow-up tests and to assess the device.
Dr. Machado and researcher Kenneth Baker, PhD, published data in Nature Medicine that showed using DBS to target the dentate nucleus, which regulates fine-control of voluntary movements and cognition, is safe and feasible.
Nine of the 12 patients showed improvements in both motor impairment and function. Additionally, Joe demonstrated substantial advancement that nearly tripled what most patients experience by undergoing routine physical rehabilitation.
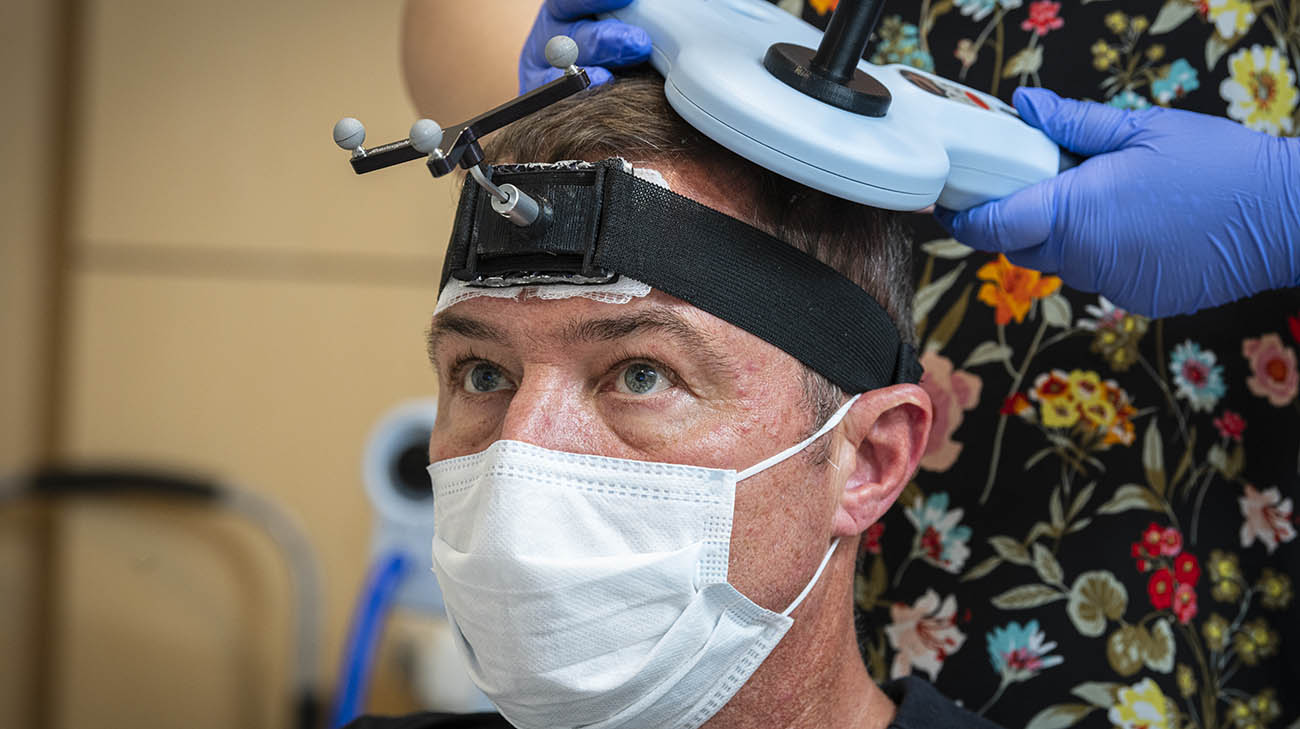
Joe says he still has a road ahead, but is grateful for the tasks he can accomplish now he never dreamed he'd be able to do. (Courtesy: Cleveland Clinic)
“These findings are reassuring for patients as the participants in the study had been disabled for more than a year and, in some cases, three years after stroke. This gives us a potential opportunity for much needed improvements in rehabilitation in the chronic phases of stroke recovery,” says Dr. Machado.
Joe, whose DBS device was surgically removed after two years of use, believes he is now 85% recovered from the initial effects of the stroke. Regarding his right hand, “I can sense when people are touching my hand, and I’m still working on developing fine motor skills for some tasks. The study did help me, and I know that what (the researchers) learned will help a lot of other people in the future.”
Related Institutes: Neurological Institute

