A stem cell transplant can treat — and sometimes cure — certain blood disorders, cancers and autoimmune diseases. The procedure replaces unhealthy stem cells with healthy ones. Your provider might get healthy stem cells from your own bone marrow. Or they might use donated stem cells from another person.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
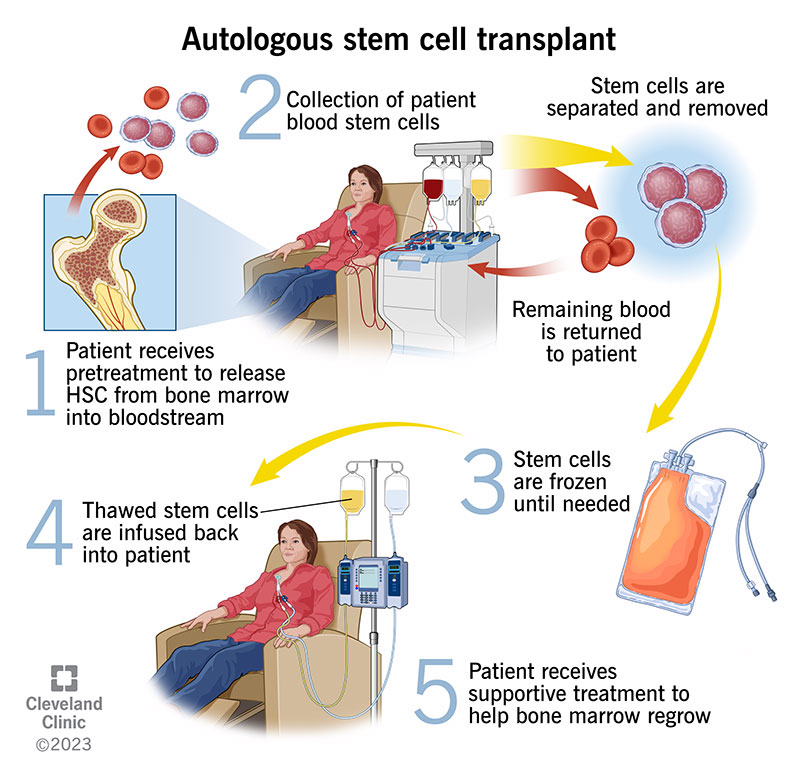
Image content: This image is available to view online.
View image online (https://my.clevelandclinic.org/-/scassets/Images/org/health/articles/autologous-stem-cell-transplant)
Stem cells create blood in your bone marrow. Sometimes, healthcare providers collect them so they can give them back to you after chemotherapy (autologous stem cell transplant). In other instances, providers collect them from a donor and give them to someone who has cancerous, abnormal or damaged stem cells (allogeneic stem cell transplant). Autologous stem cell transplants are used to treat certain kinds of cancer, blood disorders and autoimmune diseases.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Most transplants use peripheral blood stem cells (immature stem cells in your bloodstream). But some use bone marrow or umbilical cord blood stem cells. If you receive a stem cell transplant, your provider may use your own healthy stem cells or donated stem cells.
Over 22,000 people living in the United States have a disease that a stem cell transplant could cure. People usually have stem cell transplant procedures when other treatments aren’t effective or if their condition comes back after treatment.
Other names for this procedure include:
There are two types:
Autologous stem cell transplants are slightly more common than allogeneic stem cell transplants.
Whether you’re receiving donated stem cells or your own stem cells, the process feels a lot like receiving medication through a central venous catheter.
First, your provider may give you fluids and medication to help prevent side effects or reduce the chance your body may reject the new stem cells. Then, they’ll start infusing stem cells.
The process may take several hours. While you’re receiving new stem cells, your provider will frequently check for signs of fever, chills and other potential side effects.
Advertisement
A stem cell transplant itself isn’t painful. But many people develop side effects like nausea, vomiting and discomfort during recovery. Medication can help manage these side effects.
It depends on several factors, including:
Because everyone is unique, calculating overall survival rates and life expectancy can be difficult. To learn more about survival rates and what they mean for you, talk to your healthcare provider.
Stem cell transplants may help cure or slow certain blood cancers or blood disorders that didn’t respond to other kinds of treatment.
The greatest risk is that you’ll go through the procedure and your transplanted stem cells can’t slow or eliminate your illness. Cancer can come back (recur) after a stem cell transplant. But it’s less likely to happen over time.
Potential complications vary based on your overall health, age and previous treatment, including pre-transplant chemotherapy (conditioning). If you’re considering a stem cell transplant, your healthcare provider will outline potential complications so you can weigh those risks against potential benefits.
Allogeneic stem cell transplants are more likely to cause:
It depends on the type of stem cell transplant you have. People who have autologous stem cell transplants usually recover in about two months.
It can take several months to up to a year to fully recover from an allogeneic stem cell transplant. Your healthcare provider may recommend you stay in or near the hospital or treatment center for the first 100 days after your procedure.
A successful stem cell transplant can cure your condition or slow its growth. But it’s not an overnight transformation. It can take a year or more for you to recover. Here are some things that can help in the meantime:
Advertisement
Tell your provider if you develop any of these symptoms while recovering from your stem cell transplant:
If you’ve been coping with cancer or a blood disease, a stem cell transplant can be a new lease on life. It can mean hope for a cure or remission when other treatments haven’t worked. But stem cell transplants come with demanding physical challenges and significant risks. Not everyone who has cancer or blood disorders is a candidate for a stem cell transplant. And unfortunately, not everyone who needs a donor finds one. If you’re considering a stem cell transplant, talk to your healthcare provider about potential risks and benefits. They’ll evaluate your situation, options and potential outcomes.
Advertisement

Sign up for our Health Essentials emails for expert guidance on nutrition, fitness, sleep, skin care and more.
Learn more about the Health Library and our editorial process.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
Cleveland Clinic’s health articles are based on evidence-backed information and review by medical professionals to ensure accuracy, reliability and up-to-date clinical standards.
Blood cancers and conditions can be complicated. Cleveland Clinic experts offer the latest therapies and personalized treatment plans.
