Definition
Catherine Frakes Vozzo, DO
Nicole Welch, MD
Carlos Romero-Marrero, MD
Kyrsten D. Fairbanks, MD
There are 3 types of liver disease related to alcohol consumption: fatty liver, alcoholic hepatitis, or cirrhosis (Table 1). Fatty liver disease occurs after acute alcohol ingestion and is generally reversible with abstinence. Fatty liver is not believed to predispose a patient to any chronic form of liver disease if abstinence or moderation is maintained. Alcoholic hepatitis is an acute form of alcohol-induced liver injury that occurs with the consumption of a large quantity of alcohol over a prolonged period. Alcoholic hepatitis can range in severity from asymptomatic derangement of biochemistries to liver failure and death. Cirrhosis involves replacement of the normal hepatic parenchyma with extensive thick bands of fibrous tissue and regenerative nodules, which results in the clinical manifestations of portal hypertension and liver failure.
Table 1. Types of Alcoholic Liver Disease
| Type | Histologic specificity for alcoholic cause | Prognosis | Reversible |
| Fatty liver | No | Excellent | Yes |
| Alcoholic hepatitis | No | Variable | Variable |
| Cirrhosis | No | Guarded | Generally no |
Epidemiology and Risk Factors
What is known about the epidemiology of liver disease has changed due to a better understanding of nonalcoholic fatty liver disease and chronic viral hepatitis.
The prevalence of alcoholic liver disease (ALD) is difficult to define because it is influenced by many factors including genetic (eg, predilection to alcohol abuse, gender) and environmental (eg, availability of alcohol, social acceptability of alcohol use, concomitant hepatotoxic insults) factors. In the United States, it is estimated that 67.3% of the population consumes alcohol and that 7.4% of the population meets the criteria for alcohol abuse. The use of alcohol varies widely throughout the world with the highest use in the U.S. and Europe. Men are more likely to develop ALD than women because men consume more alcohol. However, women are more susceptible to alcohol hepatotoxicity and have twice the relative risk of ALD and cirrhosis compared with men. Elevated body mass index is also a risk factor in ALD as well as nonalcoholic fatty liver disease.
Ethnicity and genetics are important factors related to ALD. Cirrhosis mortality is higher in men of Hispanic, Native Americans, and native Alaskans origin compared with white populations. Genetic factors such as the presence of the patatin-like phospholipase domain containing 3 (PNPLA3) gene appear to be associated with a more severe phenotype and a poor prognosis.
In general, the risk of liver disease increases with the quantity and duration of alcohol intake. The quantity of alcohol in alcoholic beverages varies by volume base on the type of beverage (Table 2).
Table 2. Alcohol Content by Type of Alcoholic Beverage
| Type | Amount, ounces | Alcohol, grams (g) |
| Beer | 12 | 14 |
| Wine | 5 | 14 |
| Liquor (80 proof) | 1.5 | 14 |
Although alcohol use is necessary for ALD, excessive alcohol use does not necessarily promote ALD. In heavy drinkers, only 1 in 5 develops alcoholic hepatitis and 1 in 4 develops cirrhosis.
Fatty liver is a universal finding among heavy drinkers and up to 40% of those with moderate alcohol intake (10-80 mg/day) also exhibit fatty liver changes. Based on an autopsy series of men, a threshold daily alcohol intake of 40 g is necessary to produce pathologic changes of alcoholic hepatitis. Consumption of more than 80 g of alcohol per day is associated with an increase in the severity of alcoholic hepatitis, but not in the overall prevalence. There is a clear dose-dependent relation between alcohol intake and the incidence of alcoholic cirrhosis. A daily intake of more than 60 g of alcohol in men and 20 g of alcohol in women significantly increases the risk of cirrhosis. In addition, daily drinking, as compared with binge drinking, appears to be more harmful.
Pathophysiology
The liver and, to a lesser extent, the gastrointestinal tract, are the main sites of alcohol metabolism. There are 2 main pathways of alcohol metabolism in the liver: alcohol dehydrogenase and cytochrome P450 (CYP) 2E1. Alcohol dehydrogenase is a hepatocyte cytosolic enzyme that converts alcohol to acetaldehyde. Acetaldehyde subsequently is metabolized to acetate via the mitochondrial enzyme acetaldehyde dehydrogenase. CYP 2E1 also converts alcohol to acetaldehyde.
Liver damage occurs through several interrelated pathways. Alcohol dehydrogenase and acetaldehyde dehydrogenase cause the reduction of nicotinamide adenine dinucleotide (NAD) to NADH (reduced form of NAD). The altered ratio of NAD/NADH promotes fatty liver through the inhibition of gluconeogenesis and fatty acid oxidation. CYP 2E1, which is upregulated in chronic alcohol use, generates free radicals through the oxidation of nicotinamide adenine dinucleotide phosphate (NADPH) to NADP. Chronic alcohol exposure also activates hepatic macrophages, which then produce tumor necrosis factor-alpha (TNF-alpha). TNF-alpha induces mitochondria to increase the production of reactive oxygen species. This oxidative stress promotes hepatocyte necrosis and apoptosis, which is exaggerated in the alcoholic who is deficient in antioxidants such as glutathione and vitamin E. Free radicals initiate lipid peroxidation, which causes inflammation and fibrosis. Inflammation is also incited by acetaldehyde that, when bound covalently to cellular proteins, forms adducts that are antigenic.
Natural History
The spectrum of ALD can include simple hepatic steatosis, acute alcoholic hepatitis, and alcoholic cirrhosis. Overlap between these diseases is possible.
Hepatic steatosis
With alcohol abstinence, morphologic changes of the fatty liver usually revert to normal. Although the short-term prognosis in patients with alcoholic steatosis is excellent, with longer follow-up it has been found that cirrhosis develops more commonly in alcohol abusers with fatty liver changes than in those with normal liver histology. Clinical risk factors that predict progression to fibrosis, cirrhosis, or both include severe steatosis, female gender, continued excessive alcohol use, the presence of megamitochondria, mixed macrovesicular-microvesicular steatosis, and perivenular fibrosis on histology.
Alcoholic hepatitis
Scoring systems can be used to assess the severity of alcoholic hepatitis and to guide treatment. A Maddrey discriminant function (DF) score greater than 32 or a model for end-stage liver disease (MELD) score greater than 21 indicates severe alcoholic hepatitis and pharmacologic treatment should be considered. Hepatic encephalopathy and ascites are seen more often in patients who succumb to alcoholic hepatitis than in patients who survive. Long-term survival in patients with alcoholic hepatitis who discontinue alcohol use is significantly longer than in patients who continue to drink. Three-year survival approaches 90% in abstainers, whereas it is less than 70% in active drinkers. Duration of survival in both groups is considerably less than that of an age-matched population.
Alcoholic cirrhosis
Cirrhosis has historically been considered an irreversible outcome following severe and prolonged liver damage. However, studies involving patients with liver disease from many distinct causes have shown convincingly that fibrosis and cirrhosis might have a component of reversibility. For patients with decompensated alcoholic cirrhosis who undergo transplantation, survival is comparable to that of patients with other causes of liver disease with a 5-year survival of approximately 70%.
Signs and Symptoms
Typically, patients with fatty liver are asymptomatic or present with nonspecific symptoms that do not suggest acute liver disease. Supporting features on physical examination include an enlarged and smooth, but rarely tender liver. In the absence of a superimposed hepatic process, stigmata of chronic liver disease such as spider angiomas, ascites, or asterixis are likely absent.
Alcoholic hepatitis is a syndrome with a spectrum of severity thus manifesting symptoms vary. Symptoms may be nonspecific and mild and include anorexia and weight loss, abdominal pain and distention, or nausea and vomiting. Alternatively, more severe and specific symptoms can include encephalopathy and hepatic failure. Physical findings include hepatomegaly, jaundice, ascites, spider angiomas, fever, and encephalopathy.
Established alcoholic cirrhosis can manifest with decompensation without a preceding history of fatty liver or alcoholic hepatitis. Alternatively, alcoholic cirrhosis may be diagnosed concurrently with acute alcoholic hepatitis. The symptoms and signs of alcoholic cirrhosis do not help to differentiate it from other causes of cirrhosis. Patients may present with jaundice, pruritus, abnormal laboratory findings (eg, thrombocytopenia, hypoalbuminemia, coagulopathy), or complications of portal hypertension, such as variceal bleeding, ascites, or hepatic encephalopathy.
Diagnosis
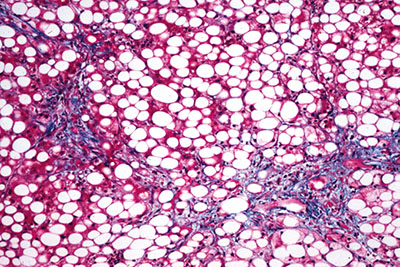
Figure 1. Histologic findings in alcoholic fatty liver disease include fat accumulation in hepatocytes.
Fatty liver is usually diagnosed in the asymptomatic patient who is undergoing evaluation for abnormal liver function tests; typically, aminotransferase levels are less than twice the upper limit of normal. No laboratory test is diagnostic of fatty liver. Characteristic ultrasonographic findings include a hyperechoic liver with or without hepatomegaly. Computed tomography (CT) and magnetic resonance imaging (MRI) can readily detect cirrhosis. On MRI, special features may be present with ALD including increased size of the caudate lobe, more frequent visualize of the right hepatic notch, and larger regenerative nodules. Liver biopsy is rarely needed to diagnose fatty liver in the appropriate clinical setting, but it may be useful in excluding steatohepatitis or fibrosis.
Typical histologic findings of fatty liver include fat accumulation in hepatocytes that is often macrovesicular, but it is occasionally microvesicular (Figure 1). The centrilobular region of the hepatic acinus is most commonly affected. In severe fatty liver however, fat is distributed throughout the acinus.Fatty liver is not specific to alcohol ingestion; it is associated with obesity, insulin resistance, hyperlipidemia, malnutrition, and various medications. Attribution of fatty liver to alcohol use therefore requires a detailed and accurate patient history.
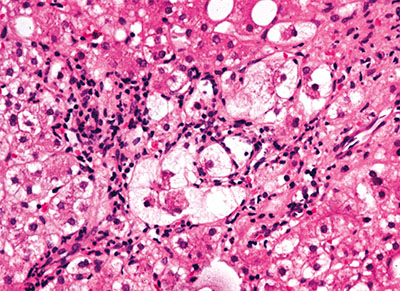
Figure 2. Inflammation and necrosis in the centilobular region of the hepatic acinus.
The diagnosis of alcoholic hepatitis is also based on a thorough history, physical examination, and review of laboratory tests. A recent consensus statement from the Alcoholic Hepatitis Consortium provided a working definition of alcoholic hepatitis that includes jaundice within 60 days of heavy consumption (> 50 g/day) of alcohol for a minimum of 6 months, serum bilirubin > 3 mg/dL, elevated AST:ALT ratio > 1.5 and no other obvious cause for hepatitis. The statement proposed classifying patients with alcoholic hepatitis as definite when a liver biopsy was used to establish diagnosis, probable when the clinical and laboratory features were present without potential confounding problems, and possible when confounding problems were present. Other common and nonspecific laboratory abnormalities include anemia and leukocytosis. In our center, considering the limited treatment options available, we routinely performed a liver biopsy via the transjugular approach to secure the diagnosis.
The classic histologic features of alcoholic hepatitis include inflammation and necrosis, which are most prominent in the centrilobular region of the hepatic acinus(Figure 2). Hepatocytes are classically ballooned, which causes compression of the sinusoid and reversible portal hypertension. The inflammatory cell infiltrate, located primarily in the sinusoids and close to necrotic hepatocytes, consists of polymorphonuclear cells and mononuclear cells. In addition to inflammation and necrosis, many patients with alcoholic hepatitis have fatty infiltration and Mallory bodies, which are intracellular perinuclear aggregations of intermediate filaments that are eosinophilic on hematoxylin-eosin staining. Neither fatty infiltration nor Mallory bodies are specific for alcoholic hepatitis or necessary for the diagnosis.
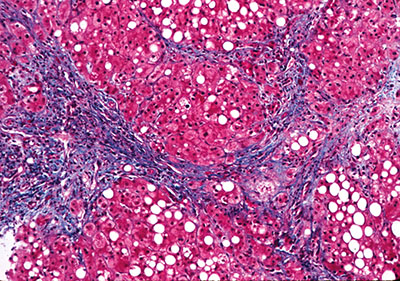
Figure 3. Histology of end-stage alcoholic cirrhosis.
The diagnosis of alcoholic cirrhosis rests on finding the classic signs and symptoms of end-stage liver disease in a patient with a history of significant alcohol intake. Patients tend to underreport their alcohol consumption, and discussions with family members and close friends can provide a more accurate estimation of alcohol intake.
Patients can present with any or all complications of portal hypertension, including ascites, variceal bleeding, and hepatic encephalopathy. The histology of end-stage alcoholic cirrhosis, in the absence of acute alcoholic hepatitis, resembles that of advanced liver disease from many other causes, without any distinct pathologic findings (Figure 3).
The overall clinical diagnosis of alcoholic liver disease, using a combination of physical findings, laboratory values, and clinical acumen, is relatively accurate (Table 3). However, liver biopsy can be justified in selected cases, especially when the diagnosis is in question. A clinical suspicion of alcoholic hepatitis may be inaccurate in up to 30% of patients. In addition to confirming the diagnosis, liver biopsy is also useful for ruling out other unsuspected causes of liver disease, better characterizing the extent of the damage, providing prognosis, and guiding therapeutic decision making.
Table 3. Physical Examination and Laboratory Findings In Alcoholic Liver Disease
| Physical Examination | |
| Constitutional |
|
| Skin |
|
| Musculoskeletal |
|
| Genitourinary |
|
| Abdomen |
|
| Neurologic |
|
| Laboratory Findings | |
| Liver synthetic function |
|
| Liver enzyme levels |
|
| Hematologic |
|
| Metabolic |
|
Treatment
The foundation of therapy for ALD is abstinence. Patients are often unable to achieve complete and durable alcohol abstinence without assistance and referral to a chemical dependency team is appropriate. Hospitalization is indicated to expedite a diagnostic evaluation of patients with jaundice, encephalopathy, or ascites of unknown cause. In addition, patients with known ALD who present with renal failure, fever, inadequate oral intake to maintain hydration, or rapidly deteriorating liver function, as demonstrated by progressive encephalopathy or coagulopathy, should be hospitalized. Patients with suspected alcoholic hepatitis should have cultures of blood, urine, and ascites to determine the presence of bacterial infections regardless of whether they have fever. The presence of systemic inflammatory response syndrome on admission is associated with a high risk of multi-organ failure syndrome, which is associated with very high mortality rate.
Nutritional Support
Supportive care for all patients includes adequate nutrition. Almost all patients with alcoholic hepatitis have some degree of malnutrition, but estimating the severity of malnutrition remains a challenge because sensitive and specific clinical or laboratory parameters are lacking. The nutritionist plays a valuable role in assessing the degree of malnutrition and guiding nutritional supplementation in malnourished alcoholic patients. The degree of malnutrition correlates directly with short-term (1-month) and long-term (1-year) mortality. At 1 year from the time of diagnosis of alcoholic hepatitis, patients with mild malnutrition have a 14% mortality rate, compared with a 76% mortality rate in those with severe malnutrition. Attempts to correct protein-calorie malnutrition with supplemental oral or parenteral nutrition, or both, have met with mixed results. In general, enteral nutrition is preferable over parenteral supplementation, and protein should be supplied to provide positive nitrogen balance. Branched-chain amino acids are useful as a supplement to maintain positive nitrogen balance in patients who do not tolerate liberal protein intake because of the development of encephalopathy; however, the expense limits routine use in all alcoholic malnourished patients. Nutritional supplementation is generally associated with an improvement in liver test results, but only rarely with a mortality benefit. Refer to the most recent practice guidelines for a summary of recommendations for daily feeding in patients with ALD. Current guidelines suggest a regular diet with 1 to 1.5 g protein and 30 to 40 kcal/kg body weight for adequate recovery. If the patient is unable to eat because of anorexia or altered mental status, a feeding tube should be considered for enteral feeding. Parenteral nutrition alone is inadequate.
Medical Treatment
The use of corticosteroids as specific therapy for alcoholic hepatitis has generated a good deal of interest. The rationale behind this approach is the possible role of the immune system in initiating and perpetuating hepatic damage. There have been more than 20 randomized controlled trials conducted in patients with severe alcoholic hepatitis treated with glucocorticoids. Some trials have failed to show a significant benefit with glucocorticoids, whereas other have demonstrated survival benefit. A meta-analysis performed in 2008 of 15 trials concluded there was no benefit from glucocorticoids, except possibly in the group with DF > 32. This was supported by a 2011 meta-analysis of the 5 largest studies, which showed a mortality benefit with prednisolone treatment.
Pentoxifylline, an oral phosphodiesterase inhibitor, is also an inhibitor of TNF synthesis. Elevated TNF levels have been associated with higher mortality from alcoholic hepatitis. A randomized, double-blind, controlled trial investigated the effects of treatment with pentoxifylline on short-term survival and progression to the hepatorenal syndrome in patients with severe alcoholic hepatitis. In patients treated with pentoxifylline, 24% (12 of 49) died compared with 52% (24 of 52) of patients receiving placebo (P= .037). Hepatorenal syndrome was the cause of death in 50% (6 of 12) of patients treated with pentoxifylline compared with 92% (22 of 24) of patients on placebo (P= .009). Subsequent meta-analysis have failed to show benefit.
The Steroids or Pentoxifylline for Alcoholic Hepatitis (STOPAH) Trial is a randomized control trial published in 2015 and the largest trial to date in alcoholic hepatitis. This study included 1,092 patients with alcoholic hepatitis and showed that neither prednisolone nor pentoxyfylline reduces all-cause mortality at 28 days. Prednisolone was associated with a non-significant mortality benefit at 28 days but no benefit at 90 days or 1 year. A meta-analysis including the STOPAH Trial showed a moderate-quality evidence that glucocorticoids alone or in combination with PTX or the antioxidant N-acetylcysteine reduced 28-day, but not 90-day mortality in patients with alcoholic hepatitis.
Patients with DF ≥ 32 or MELD score ≥ 21 should be considered for clinical trial enrollment if available. If a clinical trial is not available, a trial of glucocorticoid treatment is reasonable. The Lille score is designed to determine whether patients treated with corticosteroids should stop treatment after 1 week of treatment due to lack of treatment response. It is a good predictor of 6 months mortality and those with a score of less than 0.45 are considered to have a good prognosis and treatment with corticosteroids should be continued. Based on recent data, treatment with pentoxifylline is not supported.
Granulocyte-colony stimulating factor has been proposed as an agent to stimulate liver regeneration in patients with alcoholic hepatitis by promoting migration of bone marrow derived stem cells into the liver. A single center study from India showed a survival benefit in patients treated with granulocyte-colony stimulating factor at 90 days. Its use in patients with alcoholic hepatitis is however experimental. Other therapies that have been investigated in the treatment of alcoholic hepatitis but not found to be beneficial include propylthiouracil; infliximab; insulin and glucagon; calcium channel blockers; antioxidants such as vitamin E, S-adenosyl-L-methionine, or silymarin, which is the active ingredient in milk thistle.
Liver Transplantation
Treatment of the patient with alcoholic cirrhosis mirrors the care of patients with any other type of cirrhosis, and includes prevention and management of ascites, spontaneous bacterial peritonitis, variceal bleeding, encephalopathy, malnutrition, and hepatocellular carcinoma. Once advanced cirrhosis has occurred with evidence of decompensation (ascites, spontaneous bacterial peritonitis, hepatic encephalopathy, variceal bleeding), the patient should be referred to a transplantation center.
For more than a decade, alcoholic cirrhosis has been the second leading indication for liver transplantation in the U.S. The applicability of liver transplant for patients with severe alcoholic hepatitis is ethically controversial considering the scarcity of organs for liver transplant and the approximately 20% liver transplant waiting list mortality. Most transplantation centers require 6-months of sobriety prior to be considered for transplantation. This requirement theoretically has a dual advantage of predicting long-term sobriety and allowing recovery of liver function from acute alcoholic hepatitis. This rule proves disadvantageous to those with severe alcoholic hepatitis because 70% to 80% may die within that period. Mathurin et al found that early liver transplant in patients with severe alcoholic hepatitis versus those who were not transplanted had higher 6-month survival, and this survival benefit was maintained through 2 years of follow-up. Relapse after transplantation appears to be no more frequent than it is in patients with alcoholic cirrhosis who do not have alcoholic hepatitis.
Table 4. Treatments Investigated for Alcoholic Liver Disease
| Treatment | Routine Use Recommended | Potential Benefit |
| Abstinence | Yes | Survival |
| Nutritional support | Yes | Survival, laboratory |
| Corticosteroids | Yes | Survival |
| Pentoxifylline | No | Survival, less renal failure |
| Propylthiouracil | No | No |
| Infliximab | No | No |
| Colchicine | No | No |
| Insulin, glucagon | No | No |
| Calcium channel blocker | No | No |
| Vitamin E | No | No |
| S-adenosyl-L-methionine | No | No |
| Silymarin (milk thistle) | No | No |
| Liver transplantation | Consider (for decompensated cirrhosis) | 5-year Survival ~ 70% |
Prevention and Screening
As emphasized in the most recent national practice guidelines, health care providers must be attentive for signs of covert alcohol abuse.18 Many patients do not openly disclose an accurate history of alcohol use. In addition, no physical examination finding or laboratory abnormality is specific for ALD. All patients should therefore be screened for alcohol abuse or dependency. Abuse is defined as harmful use of alcohol with the development of negative health or social consequences. Dependency is defined by physical tolerance and symptoms of withdrawal. The CAGE questionnaire (cutting down on drinking, annoyance at others’ concerns about drinking, feeling guilty about drinking, using alcohol as an eye opener in the morning) is the preferred screening tool, with more than 2 positive answers providing a sensitivity of 71% and a specificity of 95% for alcohol dependency.
Conclusion
Clinicians should screen all patients for harmful patterns of alcohol use. All patients with alcohol-related liver disease should abstain from alcohol. For those with severe disease (ie, DF ≥32 or hepatic encephalopathy or both), and no contraindications to their use, steroids should be considered. Liver transplantation should be considered as a treatment option for patients with decompensated alcohol related cirrhosis and severe alcoholic hepatitis.
Summary
- All patients should be screened for alcoholic liver disease.
- Abstinence is the cornerstone of treatment of alcoholic liver disease.
- Alcoholic liver disease is a heterogeneous disease.
- The diagnosis of alcoholic liver disease requires a detailed patient history with supportive laboratory and imaging studies.
- Liver biopsy may be useful to confirm the diagnosis, rule out other diseases, and prognosticate.
- Patients with severe alcoholic hepatitis should be considered for enrollment in clinical trials.
- Corticosteroids treatment can be used in patients with DF > 32 or MELD > 21 without contraindication to glucocorticoid.
- Current evidence do not support the need of pentoxyfylline treatment.
- Patients with alcoholic cirrhosis should be evaluated for liver transplantation.
Suggested Reading
- Chacko KR, Reinus J. Spectrum of alcoholic liver disease.Clin Liver Dis2016; 20(3):419–427.
- Trépo E, Romeo S, Zucman-Rossi J, Nahon P.PNPLA3gene in liver diseases.J Hepatol2016; 65(2):399–412.
- Savolainen VT, Liesto K, Männikkö A, Penttilä A, Karhunen PJ. Alcohol consumption and alcoholic liver disease: evidence of a threshold level of effects of ethanol.Alcohol Clin Exp Res1993; 17(5):1112–1117.
- Lelbach WK. Cirrhosis in the alcoholic and its relation to the volume of alcohol abuse.Ann N Y Acad Sci1975; 252:85–105.
- Grant BF, Dufour MC, Harford TC. Epidemiology of alcoholic liver disease.Semin Liver Dis1988; 8(1):12–25.
- Stewart S, Jones D, Day CP. Alcoholic liver disease: new insights into mechanisms and preventative strategies.Trends Mol Med2001; 7(9):408–413.
- Zhou Z, Wang L, Song Z, Lambert JC, McClain CJ, Kang YJ. A critical involvement of oxidative stress in acute alcohol-induced hepatic TNF-alpha production.Am J Pathol2003; 163(3):1137–1146.
- Sørensen TI, Orholm M, Bentsen KD, Høybye G, Eghøje K, Christoffersen P. Prospective evaluation of alcohol abuse and alcoholic liver injury in men as predictors of development of cirrhosis.Lancet1984; 2(8397):241–244.
- Alexander JF, Lischner MW, Galambos JT. Natural history of alcoholic hepatitis. II. The long-term prognosis.Am J Gastroenterol1971; 56(6):515–525.
- Bonis PA, Friedman SL, Kaplan MM. Is liver fibrosis reversible?N Engl J Med2001; 344(6):452–454.
- Romano DR, Jiménez C, Rodriguez F, et al. Orthotopic liver transplantation in alcoholic liver cirrhosis.Transplant Proc1999; 31(6):2491–2493.
- Maddrey WC. Alcoholic hepatitis: clinicopathologic features and therapy.Semin Liver Dis1988; 8(1):91–102.
- Okazaki H, Ito K, Fujita T, Koike S, Takano K, Matsunaga N. Discrimination of alcoholic from virus-induced cirrhosis on MR imaging.AJR Am J Roentgenol2000; 175(6):1677–1681.
- Baptista A, Bianchi L, de Groote J, et al; review by an international group. Alcoholic liver disease: morphological manifestations.Lancet1981; 1(8222):707–711.
- Crabb DW, Bataller R, Chalasani NP, et al; NIAAA Alcoholic Hepatitis Consortia. Standard definitions and common data elements for clinical trials in patients with alcoholic hepatitis: Recommendation from the NIAAA Alcoholic Hepatitis Consortia.Gastroenterology2016; 150(4):785–790.
- Mitchell MC, Friedman LS, McClain CJ. Medical management of severe alcoholic hepatitis: expert review from the clinical practice updates committee of the AGA institute.Clin Gastroenterol Hepatol2017; 15(1):5–12.
- Finlayson ND. Clinical features of alcoholic liver disease. BaillieresClin Gastroenterol1993; 7(3):627–640.
- O'Shea RS, Dasarathy S, McCullough AJ; Practice Guideline Committee of the American Association for the Study of Liver Diseases; Practice Parameters Committee of the American College of Gastroenterology. Alcoholic liver disease.Hepatology2010; 51:307–328.
- Mendenhall CL, Tosch T, Weesner RE, et al. VA cooperative study on alcoholic hepatitis. II: Prognostic significance of protein-calorie malnutrition.Am J Clin Nutr1986; 43(2):213–218.
- Schenker S, Halff GA. Nutritional therapy in alcoholic liver disease.Semin Liver Dis1993; 13(2):196–209.
- Rambaldi A, Saconato HH, Christensen E, Thorlund K, Wetterslev J, Gluud C. Systematic review: glucocorticosteroids for alcoholic hepatitis—a Cochrane hepato-biliary group systematic review with meta-analyses and trial sequential analyses of randomized clinical trials.Aliment Pharmacol Ther2008; 27(12):1167–1178.
- Mathurin P, O'Grady J, Carithers RL, et al. Corticosteroids improve short-term survival in patients with severe alcoholic hepatitis: meta-analysis of individual patient data.Gut2011; 60(2):255–260.
- Hallé P, Paré P, Kaptein E, Kanel G, Redeker AG, Reynolds TB. Double-blind, controlled trial of propylthiouracil in patients with severe acute alcoholic hepatitis.Gastroenterology1982; 82(5 Pt 1):925–931.
- Akriviadis E, Botla R, Briggs W, Han S, Reynolds T, Shakil O. Pentoxifylline improves short-term survival in severe acute alcoholic hepatitis: a double-blind, placebo-controlled trial.Gastroenterology2000; 119(6):1637–1648.
- Parker R, Armstrong MJ, Corbett C, Rowe IA, Houlihan DD. Systematic review: pentoxifylline for the treatment of severe alcoholic hepatitis.Aliment Pharmacol Ther2013; 37(9):845–854.
- Thursz MR, Richardson P, Allison M, et al; STOPAH Trial. Prednisolone or pentoxifylline for alcoholic hepatitis.N Engl J Med2015; 372(17):1619–1628.
- Singh S, Murad MH, Chandar AK, et al. Comparative effectiveness of pharmacological interventions for severe alcoholic hepatitis: a systematic review and network meta-analysis.Gastroenterology2015; 149(4):958–970.e12.
- Louvet A, Naveau S, Abdelnour M, et al. The Lille model: a new tool for therapeutic strategy in patients with severe alcoholic hepatitis treated with steroids.Hepatology2007; 45(6):1348–1354.
- Singh V, Sharma AK, Narasimhan RL, Bhalla A, Sharma N, Sharma R. Granulocyte colony-stimulating factor in severe alcoholic hepatitis: a randomized pilot study.Am J Gastroenterol2014; 109(9):1417–1423.
- Rambaldi A, Gluud C. Meta-analysis of propylthiouracil for alcoholic liver disease—a Cochrane hepato-biliary group review.Liver2001; 21(6):398–404.
- Naveau S, Chollet-Martin S, Dharancy S, et al; Foie-Alcool group of the Association Française pour l’Etude du Foie. A double-blind randomized controlled trial of infliximab associated with prednisolone in acute alcoholic hepatitis.Hepatology2004; 39(5):1390–1397.
- Bird G, Lau JY, Koskinas J, Wicks C, Williams R. Insulin and glucagon infusion in acute alcoholic hepatitis: a prospective randomized controlled trial.Hepatology1991; 14(6):1097–1101.
- Bird GL, Prach AT, McMahon AD, Forrest JA, Mills PR, Danesh BJ. Randomised controlled double-blind trial of the calcium channel antagonist amlodipine in the treatment of acute alcoholic hepatitis.J Hepatol1998; 28(2):194–198.
- Trinchet JC, Balkau B, Poupon RE, et al. Treatment of severe alcoholic hepatitis by infusion of insulin and glucagon: a multicenter sequential trial.Hepatology1992; 15(1):76–81.
- Mezey E, Potter JJ, Rennie-Tankersley L, Caballeria J, Pares A. A randomized placebo controlled trial of vitamin E for alcoholic hepatitis.J Hepatol2004; 40(1):40–46.
- Rambaldi A, Gluud C. S-adenosyl-L-methionine for alcoholic liver diseases.Cochrane Database Syst Rev2006; (2):CD002235.
- Rambaldi A, Jacobs BP, Iaquinto G, Gluud C. Milk thistle for alcoholic and/or hepatitis B or C liver diseases—a systematic Cochrane hepato-biliary group review with meta-analyses of randomized clinical trials.Am J Gastroenterol2005; 100(11):2583–2591.
- Mathurin P, Moreno C, Samuel D, et al. Early liver transplantation for severe alcoholic hepatitis.N Engl J Med2011; 365(19):1790–1800.
- Addolorato G, Bataller R, Burra P, et al. Liver transplantation for alcoholic liver disease.Transplantation2016; 100(5):981–987.
- Aertgeerts B, Buntinx F, Kester A. The value of the CAGE in screening for alcohol abuse and alcohol dependence in general clinical populations: a diagnostic meta-analysis.J Clin Epidemiol2004; 57(1):30–39.