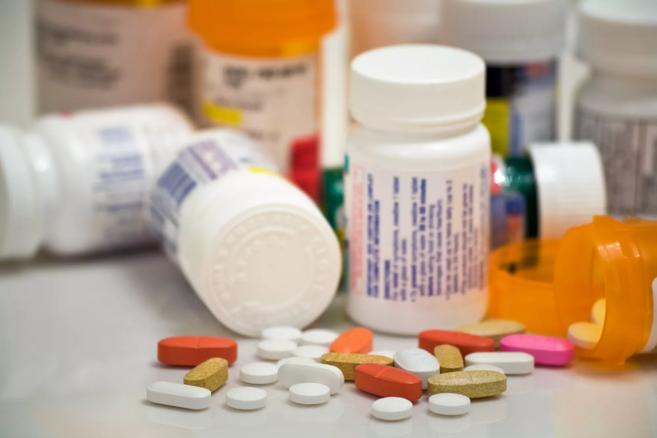
Dozens of locations will collect unused prescriptions and over-the-counter medications
Innovative program will address unique health needs of women during midlife and beyond
25 examples of progress showcase the impact of successful collaboration




Innovation, treatments and breakthrough research told through the eyes of inspiring patients and doctors.
View our storiesServices include fulfilling requests for live, remote and pre-recorded interviews, as well as providing a range of assets such as infographics, photos and b-roll footage.
Resources for journalists